Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
Given-
Mass of K2SO4, w = 25 mg = 25 X 10-3 g,
Molar mass of K2SO4 = (39*2) + (32*1) + (16*4) = 174 g mol-1
Volume V = 2 liter
T = 250C + 273 = 298 K (add 273 to convert in Kelvin)
The reaction of dissociation of K2SO4 is written as,
K2SO4 → 2K + + SO42-
Number if ions produced = 2 + 1 = 3, hence vant Hoff's factor, I = 3
Here, we use vant Hoff's equation for dilute solutions, given as,
πV = inRT
where, n is the number of moles of solute, R is solution constant which is equal to the gas constant (0.082) and T is the absolute temperature (298 K).


Hence, the osmotic pressure of a solution is 5.27x10-3atm
New answer posted
a year agoContributor-Level 10
16.24
Alitame is a high potency artificial sweetener and it is not possible to control the sweetness imparted to food by alitame. Alitame is an artificial sweetener that is 2,000 times as sweet as sugar. This sweetness is very high as compared to the natural sugar and use of such sweetener is very critical while preparing sweet dishes. The structure of alitame is as follows:
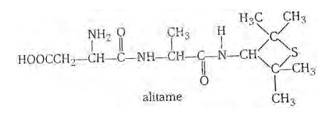
New answer posted
a year agoContributor-Level 10
Given-
Vant Hoff's factor, I = 2.47
osmotic pressure, π = 0.75 atm
Volume of solution = 2.5L.
To determine the amount of CaCl2, we use vant Hoff's equation for dilute solutions, given as,
πV = inRT
where, n is the number of moles of solute, R is solution constant which is equal to the gas constant and T is the absolute temperature.

Hence, the amount of CaCl2 dissolved is 3.425g
New answer posted
a year agoContributor-Level 10
Given-
KH for O2 = 3.30 * 107 mm Hg,
KH for N2 = 6.51 * 107 mm Hg
Percentage of oxygen (O2) = 20 %
Percentage of nitrogen (N2) = 79%
Total pressure = 10 atm
Using Henry's law,

where, p is the partial pressure of gas in the solution and KH is Henry's constant.

Thus, the mole fraction of oxygen in solution, xoxy = 4.61x10-5
and the mole fraction of nitrogen in solution, xnit is 9.22x10-5
New answer posted
a year agoContributor-Level 10
16.23
Artificial sweetening agents like Saccharin, Alitame, Sucrolose can be used in the preparation of sweets for a diabetic patient as they do not add any calories to the body.
Diabetic people are advised to consume low-calorie diet [fewer carbohydrates & more proteinaceous and fiber rich]. The refined sugar like sucrose adds calorie to the diet but complex sugar and starches do not add to the calorie intake of a person and at the same time impart a sweet taste to diet. Artificial sweeteners are generally either complex sugar or protein in nature
New answer posted
a year agoNew answer posted
a year agoContributor-Level 10
16.22
Artificial sweetening agents refer to those compounds which impart sweet taste to any food product but at the same, they do not add any calories to the body. Example: Aspartame, Alitame, Saccharin etc.
New answer posted
a year agoContributor-Level 10
The Ptotal for the values given in the graph is found out and plotted in the graph.
ptotal (mm Hg) | 632.8 | 603.0 | 579.5 | 562.1 | 580.4 | 599.5 | 615.3 | 641.8 |

It can be observed from the graph that the plot for the p total of the solution curves downwards. Therefore, the solution shows negative deviation from the ideal behaviour.
New answer posted
a year agoContributor-Level 10
16.21
At elevated temperatures, aspartame is unstable and break down to give a tasteless compound because of which aspartame is limited to cold foods and drinks.
Aspartame is methyl ester of the dipeptide obtained from phenylalanine and aspartic acid. It is about 180 times as sweet as cane sugar. The structure of aspartame is as follows:
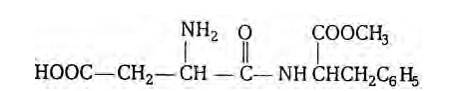
New answer posted
a year agoContributor-Level 10
Given-
Mass of liquid A, WA = 100g, Molar mass, MA = 140 g mol-1
Mass of liquid B, WB = 1000 g, Molar mass, MB = 180 g mol-1
Using the formula below calculate the no. of moles in liquid A and B.
Number of moles = Mass / Molar Mass
Number of moles of liquid A, MA = 100/140 = 0.714 mol-1
Number of moles of liquid B, MB = 1000/ 180 = 5.556 mol-1
Using the formula,
mole fraction of a liquid = No. of moles of the liquid / total no of moles
we calculate the mole fraction of liquids A and B.
→ Mole fraction of A,
xA = 0.714 / (0.714 + 5.556)
∴ xA = 0.114
→ Mole fraction of B,
xB = 1- xA = 1 - 0.114
∴ xB = 0.886
Vapour pressure of pure liquid B, Po
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers




