Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
16.8
In medicinal chemistry, drug targets refer to the specific key molecules involved in certain metabolic pathways that result in specific diseases. Carbohydrates, proteins, lipids, and nucleic acids are examples of drug targets.
Drugs are chemical agents designed to inhibit these target molecules by binding with the active sites of the key molecules.
New answer posted
a year agoContributor-Level 10
16.6
The basis on which drugs are classified in different ways is : - of drugs and the reasons for classification are as follows:
(i) On the basis of pharmacological effect:
This classification provides doctors the whole range of drugs available for the treatment of a particular type of problem. Hence, such a classification is very useful to doctors.
(ii) On the basis of drug action:
This classification is based on the action of a drug on a particular biochemical process.
(iii) On the basis of chemical structure:
This classification provides the range of drugs sharing common structural features and often having similar pharmaco
New question posted
a year agoNew answer posted
a year agoContributor-Level 10
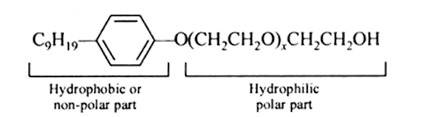
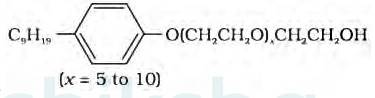
The functional group present in the molecule is:
1. Ether
2. Primary Alcoholic group
Alcohol group can recognize by the presence of –OH group present in the given organic compound and on the other hand, the ether group can be identified by the presence of R-O-R bonds where R represents the alkyl group present in the compound.
New question posted
a year agoNew answer posted
a year agoContributor-Level 10
16.4
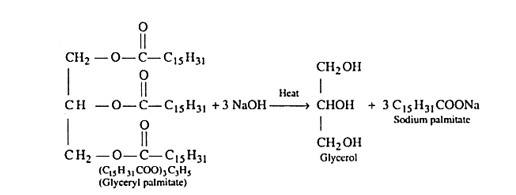
Soaps are metallic salts of higher fatty acids such as palmitic acid, stearic acid, oleic acid etc. The process of saponification involves the hydrolysis of an oil or fat with an alkali (sodium hydroxide or potassium hydroxide). Oils and fats are glyceryl esters of fatty acids and may be regarded as mixed glycerides. Sodium soaps are prepared by heating the fats by sodium hydroxide.
(i) (C15H31COO)3C3H5 – Glyceryl palmitate
(ii) (C17H32COO)3C3H5 – Glyceryl oleate
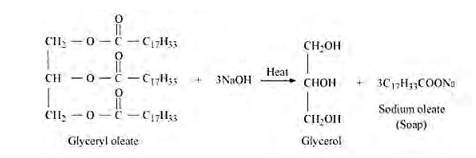
New question posted
a year agoNew answer posted
a year agoContributor-Level 10
16.3
Diabetes mellitus is the third leading cause of death in the world. It is a medical condition in which the glucose is not consumed in the body due to inefficiency in the secretion of insulin [hormone which converts glucose to glycogen and store in liver]. Diabetes is the precursor for many other diseases like obesity, heart attacks, stroke etc.
Therefore Diabetic people are advised to consume low-calorie diet [fewer carbohydrates & more proteinaceous and fiber rich]. The refined sugar like sucrose adds calorie to the diet but complex sugar and starches do not add to the calorie intake of a person and at the same time impart a
New answer posted
a year agoContributor-Level 10
Given:
Molar mass of non-volatile solute = 40g
Let no. of moles of solute be n.
Mass of octane = 114g
Molar mass of octane (C8H18) = 12 * 8 + 1 * 18 = 114g/mol
Moles of octane = given mass/molar mass
⇒ n = 114/114 moles
⇒ n = 1 mole
Molar fraction of solute,
x2 = moles of solute / moles of solute + moles of octane
⇒ x2 = n/n + 1
Let the vapour pressure of original solvent (without solute) be p1?
Accordingly after addition of solute vapour pressure of solution reduces to 80% i.e.
0.8 p1? = p1
Applying the formula:

⇒ n/n + 1 = 0.2
⇒ 0.2n + 0.2 = n
⇒ n = 0.25 moles
Hence, mass of solute is:
moles = given mass/molar mass
⇒ 0.25moles = ma
New answer posted
a year agoContributor-Level 10
Given: 1 molal solution means 1 mole of solute present in 1000g of water solvent)
Molecular weight of water = H2O = 1 * 2 + 16 = 18g/mol
No. of moles of water, n = given mass /molecular weight
⇒ n = 1000/18 = 55.56 gmol-1
Mole fraction of solute in solution, x2 = moles of solute/ (moles of solute + moles of water)
⇒ x2 = 1/ (1 + 55.56)
⇒ x2 = 0.0177
Given vapour pressure of pure water at 300k is 12.3 kpa
Apply the formula:
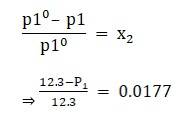
⇒ P1 = 12.0823kpa
which is the vapour pressure of the solution.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers