Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
Molar mass of Nalorphene = 311g/mol
Now 1000g of solution contains 1.5 * 10-3 moles of Nalorphene (Molality of solution = moles of solute/mass of solvent (in kg)
⇒ 1.5 * 10-3 moles of Nalorphene = 1.5 * 10-3 * 311 = 0.4665g of Nalorphene
Therefore, total mass of the solution = (1000 + 0.4665) g
⇒ total mass = 1000.4665 g
This implies that the mass of the solution containing 0.4665 g of nalorphene is 1000.4665 g.
Therefore, mass of the solution containing 1.5 mg of nalorphene is:
Mass = 1000.4665 X 1.5 X 10-3 / 4.665 g
⇒ mass of solution containing required ions = 3.22 g
Hence, the mass of aqueous solution required is 3.22 g.
New answer posted
a year agoContributor-Level 10
16.16
Magnesium or Aluminium hydroxides do not tackle with the root cause of acidity that is they reduce the acidity by reacting with excess acid present in the stomach [neutralization reaction].
On the other hand cimetidine and ranitidine relieve acidity by preventing interaction of histamine with receptors of stomach walls and hence release of excess acid by stomach walls is stopped. So the root cause of acidity is totally eliminated.
Therefore cimetidine and ranitidine relieve the acidity by tackling the root cause whereas magnesium or aluminum hydroxide just relieves by healing only the mere cause of acidity.
New answer posted
a year agoContributor-Level 10
Total mass of solution = 6.5g + 450g = 456.5g
Therefore mass percentage of aspirin in solution = (mass of aspirine/total mass of solution) = 6.5/456.5
⇒ mass % of aspirine = 1.424%
New answer posted
a year agoContributor-Level 10
16.15
Antiseptics and disinfectants are effective against micro-organisms. Antiseptics are applied to the living tissues such as wounds, cuts, ulcers, and diseased skin surfaces, while disinfectants are applied only to inanimate objects such as floors, drainage system, instruments, etc. Disinfectants are harmful to the living tissues. Iodine is an example of a strong antiseptic. Tincture of iodine (2 − 3 percent of solution of iodine in alcohol − water mixture) is applied to wounds. 1 percent solution of phenol is used as a disinfectant.
New answer posted
a year agoContributor-Level 10
The Solubility product of CuS (ksp) = 6 * 10-16
CuS → Cu ++ + S2-
Let the s be solubility of CuS in mol/L
Ksp = [ Cu ++ ] [S2]
Ksp = solubility product
6 * 10-16 = s * s = s2
⇒ S = 2.45 * 10-8 mol/L
Hence, the maximum molarity of CuS in an aqueous solution is 2.45 * 10-8 mol/L
New answer posted
a year agoContributor-Level 10
16.14
Antibiotics that are effective against a wide range of gram-positive and gram-negative bacteria are known as broad spectrum antibiotics. Chloramphenicol is an example of a broad spectrum antibiotic. It has the following structure: -
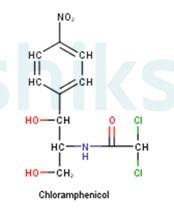
It can be used for the treatment of typhoid, dysentery, acute fever, pneumonia, meningitis, and certain forms of urinary infections. Two other examples of broad spectrum antibiotics are vancomycin and ofloxacin. Ampicillin and amoxicillin? synthetically modified from penicillin are also broad spectrum antibiotics.
New answer posted
a year agoContributor-Level 10
Mass of ions = 92g
Molar mass of ions = Na+ = 23g (neglect the mass lost due to absence of a electron)
Moles of ions = mass of ions/molar mass
⇒ n = 92/23 moles
⇒ n = 4moles
Molality of solution = moles of solute/mass of solvent (in kg) Molality = 4/1 = 4M
New answer posted
a year agoContributor-Level 10
16.13
Anti-depressant drugs are needed to counteract the effect of depression. These drugs function by inhibiting the enzymes catalysing the degradation of the neurotransmitter, noradrenaline. As a result, the important neurotransmitter is slowly metabolised and then it can activate its receptor for longer periods of time.
Two anti-depressant drugs are:
(i) Iproniazid
(ii) Phenelzine
New answer posted
a year agoContributor-Level 10
- Water is a polar compound (due to electronegativity difference between O and H) . We know that “like dissolves like”. So, a non-polar compound will be more soluble in non-polar solvent as compared to polar compound.
- Phenol has the polar group -OH and non-polar group –C6H5 and it can not form H bonding with water (presence of bulky non-polar group) . Thus, phenol is partially soluble in water
- Toluene has no polar Thus, toluene is insoluble in water.
- Formic acid (HCOOH) has the polar group -OH and can form H-bond with water. Thus, formic acid is highly soluble in water
- Ethylene glycol (OH-CH2-CH2-OH) has polar -OH group and can form H-
New answer posted
a year agoContributor-Level 10
16.12
Specific drugs affect only some specific or particular receptors. Antacids and anti-allergic drugs work on different receptors. This is the reason why antacids and anti-allergic drugs do not interfere with each other's functions, but interfere with the functions of histamines.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers
