Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
Given:
Molarity of HCl, = 0.1 M
Mass of Mixture = 1g
To find: Volume of HCl to react completely with mixture
Formula:
Molarity, Mo = number of moles of solute/ Volume of solution in litres
Solution:
Calculation of Amount of each component in mixture:
⇒ Let the amount of Na2CO3 be X g
⇒ And Let amount of NaHCO3 be [1-X] g
⇒ Molecular Weight of Na2CO3 = [23 * 2] + [12] + [3 * 16]
= 106 g
⇒ Molecular Weight of NaHCO3 = [23] + [1] + [12] + [3 * 16]
= 84 g
⇒ Number of moles of NaHCO3 = 1-x / 84
⇒ Number of moles of Na2CO3 = x / 106
Now it is given in the question that the mixture is equimolar, so
⇒ Number moles of Na2CO3 = Number of moles
New answer posted
a year agoContributor-Level 10
Given:
Concentration of Nitric Acid, HNO3 = 68%
Density of solution, d = 1.504 g/ml
To find: Molarity, Mo
Formula:
Density, d = Mass (M) / volume (V)
Molarity, Mo = Number of moles of solute/ Volume of solution in litres
Solution:
68% of Nitric acid by mass in aqueous solution means that 68g [68 * 100]/100] of Nitric acid present in 100g of solution.
⇒ Molecular mass of Nitric Acid, HNO3 = [1 * 1] + [1 * 14] + [16 * 3]
= 63g
⇒ Number of moles of Nitric Acid = [68/63]
= 1.079 moles
⇒ Given Density, d = 1.504 g/ml
⇒ Volume, v = [100/1.504]
= 66.489 ml
⇒ Molarity, Mo = [1.079/66.489] * 1000
= 16.23 M
Therefore the molarity of the sample is
New answer posted
a year agoContributor-Level 10
As the name signifies, a solid solution is one in which solvent is solid.So considering this aspect absorption of hydrogen over platinum or palladium is an example of such solution. Platinum or palladium is used as a catalyst in hydrogenation processes.
New question posted
a year agoNew answer posted
a year agoContributor-Level 10
A solution is a homogeneous mixture of two or more than two substances on molecular level whose composition can vary within certain limits.
The part or component of the mixture present in a lesser amount is called
the SOLUTE and the one present in larger amount is called the SOLVENT. For eg- small amount of salt [solute] dissolved in water [solvent].
There are nine types of solutions formed. They are:
Sr.No. |
State of solute |
State of solvent |
Examples |
1 |
GAS |
GAS |
Air |
2 |
GAS |
LIQUID |
Oxygen in water, carbonated water |
3 |
SOLID |
GAS |
Smoke particles in air, dust particles in air |
4 |
LIQUID |
GAS |
Mist |
5 |
LIQUID |
LIQUID |
Alcohol in water |
6 |
LIQUID |
SOLID |
Mercury in silver |
7 |
GAS |
SOLID |
Adsorption of hydrogen over palladium or platinum |
8 |
SOLID |
LIQUID |
Sugar in water |
9 |
SOLID |
SOLID |
Carbon in Iron(steel), Alloy |
Out of these nine types solution, solid in liquid, liquid in liquid & gas in liquid are very common. When the components of the solution are mixed, the resulting solution may exist in any of the three possible states of matter that is solid, liquid or gaseous. They are: (1) Gaseous solution: In such solutions solvent is Since the solvent is gas,the solute can be solid, liquid or gas. For example, a mixture of oxygen and nitrogen gas is a gaseous solution. (2) Liquid solution: In such type of solutions liquid acts as the solvent. The solute in these solutions may be gas, liquid, or solid. (3) Solid solutions: As the name suggests, in such solutions solid acts as the solvent. The solute in these solutions may be a gas, liquid or solid. For example, a solution of copper in gold is a solid solution. |
New answer posted
a year agoContributor-Level 10
11.86

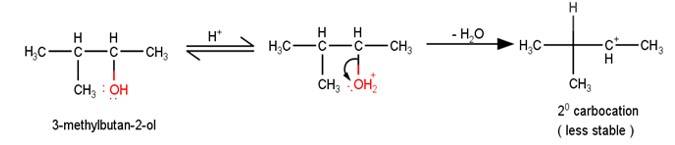
The first step in the mechanism of th e given reaction is protonation of the alcohol followed by loss of water to give a 20 carbocation.
e given reaction is protonation of the alcohol followed by loss of water to give a 20 carbocation.
2. The next step is a rearrangement of the 20 carbocations formed in the above step is less stable it rearranges by a 1,2-hydride shift to form more stable 3° carbocations.
New answer posted
a year agoContributor-Level 10
11.85
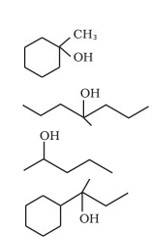
We know that the addition and elimination reactions are opposite of each other.Hence, for solving the above questions our approach should be to first dehydrate a suitable alcohol to give either a single alkene or a mixture of an alkene, if we obtain a mixture of alkene then we would have to detect which of the alkene will give us the desired alcohol. Wherever required the acid- catalyzed addition of water to alkenes will follow Markovnikov's rule.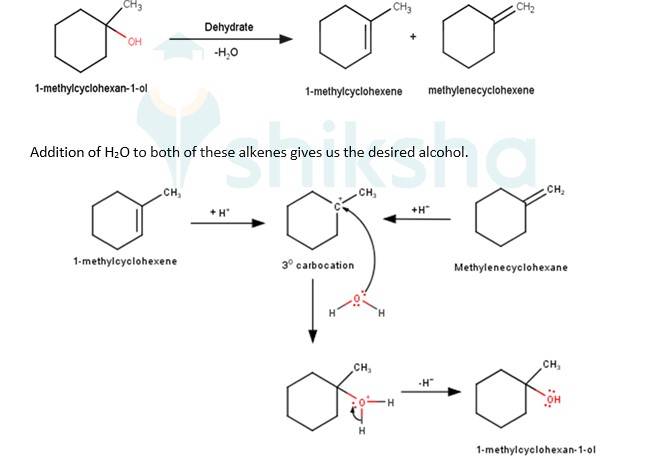
New answer posted
a year agoContributor-Level 10
11.84
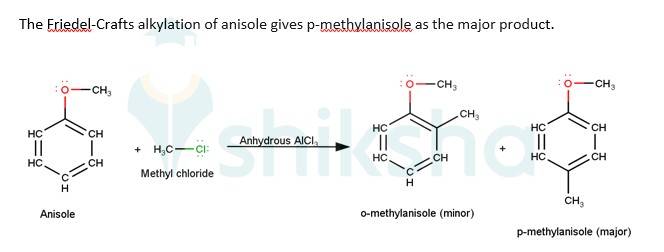
The driving force of all the reactions given to the question is that the alkoxy group is an ortho and para directing group because it exerts its +R effect in the benzene ring. Para position being comparatively more stable than the ortho position is usually preferred because ortho position leads to stearic hindrance, hence the major product is mostly the para- substituted compound.
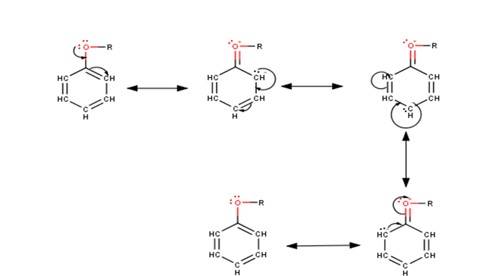
As seen from the resonating structures above the structure in which the negative charge is in the para position will form a more stable product when attacked by an electrophile. Hence in the following reactions, we will be considering that resonating stru
New answer posted
a year agoContributor-Level 10
11.83
The reaction of HI with methoxymethane yields two different sets of products depending upon the initial amount of HI taken.
When equal moles of HI and methoxymethane are taken, a mixture of methyl alcohol and methyl iodide is
The mechanism is given below:
In the first step, methoxymethane reacts with hydrogen iodide to extract a proton to give the dimethyloxonium ion.
In the second step of the reaction, the Dimethyloxonium ion reacts with the iodide ion present to yield methyl iodide and methyl alcohol as the product via SN2 pathway.
If an excess of HI is used the methyl alcohol formed in Step II is also converted into methyl iod
New answer posted
a year agoContributor-Level 10
11.82
(i) In aryl alkyl ethers the +R effect of the alkoxy group leads to an increase in the electron density of the benzene ring as they push electrons into the ring making the benzene ring activated towards electrophilic substitution reactions. This could be understood more clearly from the following resonating structures : -
(ii) It could be clearly seen from the above resonating structures that the electron density increases more at the ortho and para positions as compared to the meta positions. Hence, we can conclude that the alkoxy group directs the incoming substituents to ortho and para positions in the benzene ring.
For example
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers