Physics
Get insights from 5.6k questions on Physics, answered by students, alumni, and experts. You may also ask and answer any question you like about Physics
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoContributor-Level 10
For a conducting sphere, the electric field E = σ/ε? and potential V = σR/ε?
When two spheres are connected by a wire, their potentials become equal: V? = V?
σ? R? /ε? = σ? R? /ε?
σ? R? = σ? R? ⇒ σ? /σ? = R? /R?
New answer posted
7 months agoContributor-Level 10
The reaction is X²? → Y¹²? + Z¹²?
Binding energies per nucleon are: X=7.6 MeV, Y=8.5 MeV, Z=8.5 MeV.
Gain in binding energy (Q) = (Binding energy of products) - (Binding energy of reactants)
Q = (120 * 8.5 + 120 * 8.5) - (240 * 7.6) MeV
Q = (2 * 120 * 8.5) - (240 * 7.6) MeV = 2040 - 1824 = 216 MeV.
New question posted
7 months agoNew answer posted
7 months agoContributor-Level 10
In n-type semiconductor majority charge carriers are e- and P type semiconductor majority charge carriers are holes.
neA
New answer posted
7 months agoContributor-Level 9
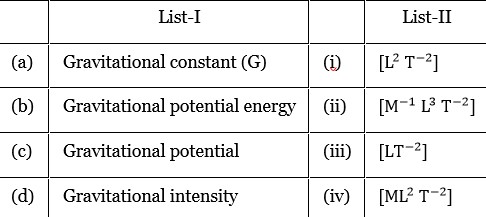
Gravitational constant
Gravitational potential energy
Gravitational potential
Gravitational intensity
New answer posted
7 months agoContributor-Level 10
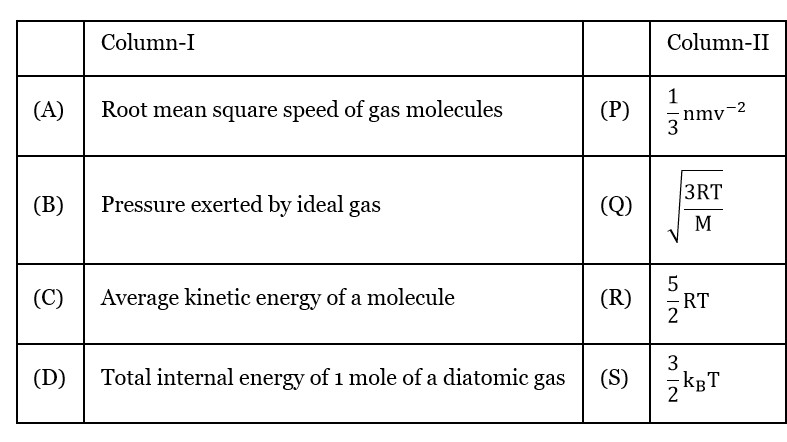
Root mean square speed of gas molecules
Pressure exerted by ideal Gas
Average kinetic energy of a molecular
Total internal energy of 1 mole of a diatomic gas
(For 1 mole diatomic gas)
New answer posted
7 months agoContributor-Level 10
Mass
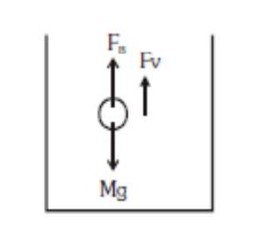
Density of ball
Density of glycerine
For constant velocity,
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers