Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoContributor-Level 10
Effective number of atom in C.C.P
= 4
Number of octahedral void = 4
Number of cations = 4
Number of anion = 4
Formula of compound = A4B4
Empirical formula = AB
Ans. x = 1
New answer posted
7 months agoContributor-Level 10
50 ml of 1 (M) HCl + 30 ml of 1 (M) NaOH
NaOH + HCl -> NaCl + H2O
30 * 1 mmol 50 * 1 mmol
0 mmol 20 mmol
x = 6021
Ans. = 6021
New answer posted
7 months agoBeginner-Level 5
Students should prepare complete syllabus when they have time to prepare. However, you can use the list of highweightage chapters in last minute revision for scoring well.
- The p-Block Elements: This chapter holds a high weightage of 8–10 marks. (in the latest syllabus this is deleted)
Aldehydes, Ketones, and Carboxylic Acids: This chapter contributes around 8–10 marks.
Biomolecules: This chapter accounts for around 8 marks.
Chemical Kinetics: This chapter holds a high weightage of 5-6 marks.
The d- and f-Block Elements: This chapter contributes around 5-6 marks.
Amines: This chapter contributes around 5-6 marks.
New answer posted
7 months agoContributor-Level 9
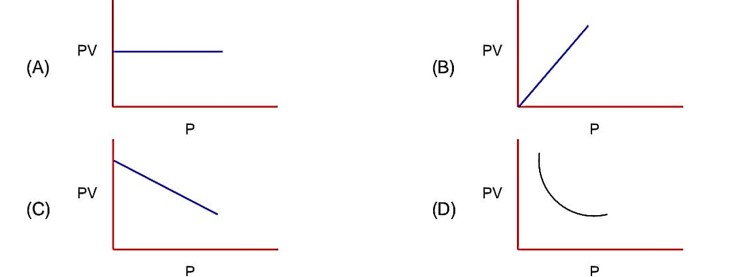
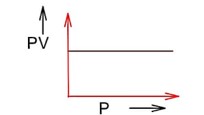
PV = nRT PV = constant (at constant T)
Pressure increases & volume decreases, PV remains constant at constant T.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers