Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoContributor-Level 10
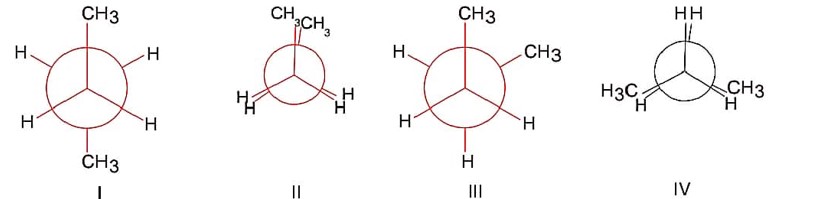
Structure (I) is anti conformer.
Structure (II) is fully eclipsed conformer.
Structure (III) is skew or gauche conformer.
Structure (IV) is partially eclipsed.
order of stability
(I) > (III) > (IV) > (II)
Order of P.E. is (II) > (IV) > (III) > (I).
New answer posted
7 months agoContributor-Level 10
Na/H2 can not reduce a functional group as Na does not behaves as catalyst here.
New answer posted
7 months agoContributor-Level 10
Stability order of oxides (X2O) is,
l2O > Cl2O > Br2O
Bonds of halogen & oxygen are covalent due to less EN difference.
Stability of (I - O) bond is higher due to less polarity and that of (Cl-O) bond is higher due to multiple bonding.
New answer posted
7 months agoContributor-Level 10
(a) Cell constant =
(b) molar conductivity
= Scm2 mol-1
(c) Conductivity
(d) degree of dissociation =
New answer posted
7 months agoNew answer posted
7 months agoContributor-Level 10
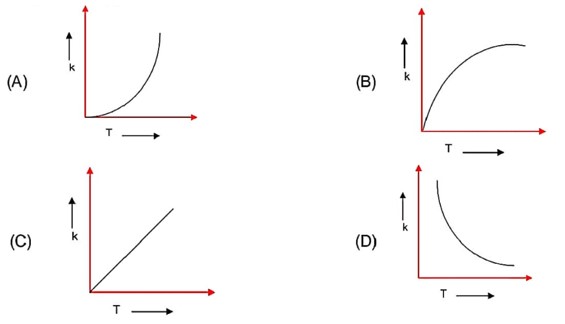
Rate constant depends on temperature as
Here; as T increases rate constant increases exponentially.
New answer posted
7 months agoContributor-Level 10
In reaction ;
Colour of Fe2+ is green and that of Fe3+ is yellow.
New answer posted
7 months agoContributor-Level 9
Co has +2 oxidation state
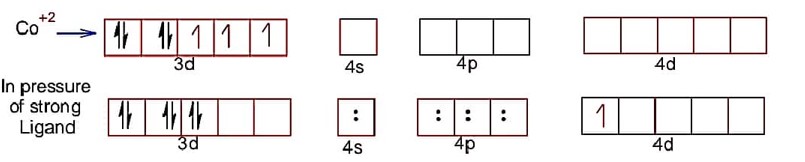
Hence, electronic configuration of CO2+ is . In complex given ligand CN is strong hence, after pairing in d-subshell, total number of unpaired electron =
spin magnetic moment =
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers