Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
Following are the
Cr2+ - 1926 KJ/mole
Mn2+ - 1860 KJ/mole
Fe2+ - 2000 KJ/mole
Co2+ - 2078 KJ/mole
New answer posted
8 months agoContributor-Level 10
(a) low solubility of LiF in H2O due to high lattice energy.
(b)
(c) Na in liq NH3 forms electronated ammonia thus conducting in nature.
(d)
New answer posted
8 months agoContributor-Level 10
In clark's method Ca (OH)2 is used for softening hard water.
Thus CaCO3 and Mg (OH)2 are formed in reaction.
New answer posted
8 months agoContributor-Level 10
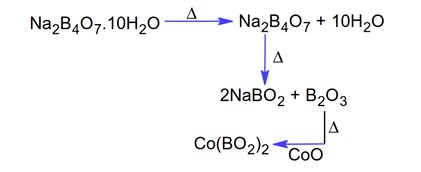
Here 'X' is the Gypsum (CaSO4.2H2O) which is used to enhance the setting of time
New answer posted
8 months agoContributor-Level 10
Liquation refining process is applicable for the metal having low m.p, but containing impurities have higher m.p
New answer posted
8 months agoBeginner-Level 5
The difference in boiling or melting point, even after nearly the same molecular geometry, is because opf the presence of hydrogen bond. HF forms intermolecular hydrogen bonds due to fluorine's high electronegativity. This intermolecular hydrogen bonding requires more energy due to break the hydrogen bonds and melt or boil. So, stronger intermolecular forces of HF result in a higher boiling point.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers