Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
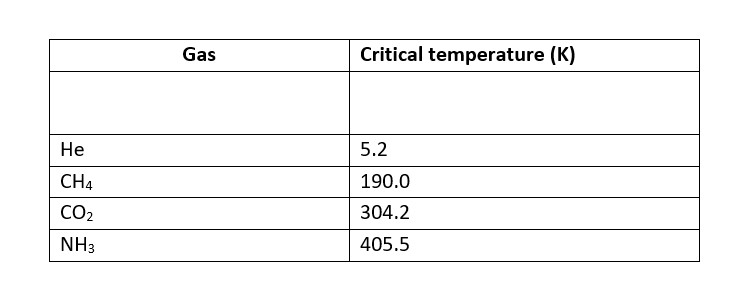
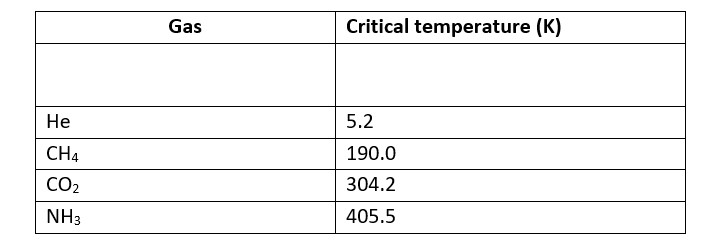
Higher adsorbtion of gas is corresponds to higher liquefaction and higher liquefaction is directly proportional to the higher critical temperature.
New question posted
8 months agoNew answer posted
8 months agoNew answer posted
8 months agoContributor-Level 10
Mass of pure carbon in coal = 0.6 * 1000gm *
Mass of carbon converted into CO = 6
Mass of carbon converted into CO2 = 360 – 216 = 144gm Þ Mole =
C (s) + O2 (g) -> CO2 (g) + 400KJ
Mole – 12 for CO2 production, Hence total energy produced = 400 * 12 = 4800KJ
Mole = 18 for CO production, Hence energy produced = 100 * 18 = 1800KJ
Total heat = 4800 + 1800 = 6600KJ
New answer posted
8 months agoContributor-Level 10
(a) Nitric oxide formation -> Pt is used as catalyst
(b) Haber's process -> Fe is used as catalyst
(c) Hydrolysis of ester -> Acid (H2SO4) is used as catalyst
(d) SO3 formation -> NO is used as catalyst
New answer posted
8 months agoContributor-Level 10
(a)
During discharging of secondary battery this reaction takes place.
(b)
Primary battery mercury cell reaction
(c)
During charging of secondary battery PbSO4 reacts and generated
(d) reacts in fuel cell to form
New answer posted
8 months agoContributor-Level 10
4 moles of HNO3 produced 3 mol of KNO3
Here mole of produced KNO3 =
If 3 mol of KNO3 produced by 4 moles of HNO3
1 mole of KNO3 produced by moles of HNO3
and mole of KNO3 produced by moles of HNO3 = 1.45 mole of HNO3
Hence mass of HNO3 = mole * mol.wt = 145 * 63 = 91.48 91.5gm
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers