Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
Let we take of solution
Mass of solute = Volume * Density
= 0.5
= 0.525 gram
Mass of solution = 1 kg. [considering very dilute solution]
Mass of solvent = 1000 – 0.525 = 999.475 gram
New answer posted
8 months agoContributor-Level 10
XeF2 sp3d2, square pyramidal
XeF6 sp3d3, Distorted octahedral
XeOF4 sp3d2, square pyramidal
New answer posted
8 months agoContributor-Level 10
Let a moles of SO2Cl2 is taken
Then no. of moles of H2SO4 = a moles
No. of moles of HCl = 2a moles
No. of moles of NaOH required = 2a + 2a = 4a = 16
New answer posted
8 months agoContributor-Level 10
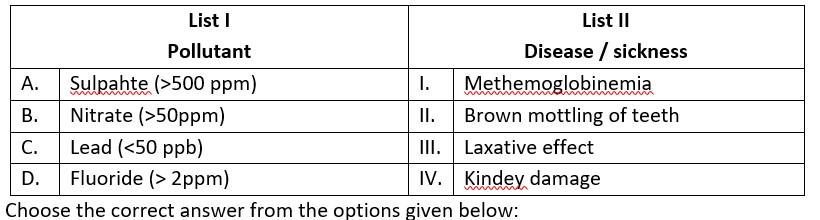
(A) Sulphate (>500 ppm) ® causes laxative effect that leads to dehydration.
(B) Nitrate (> 50 ppm) ® causes methemoglobinemia, skin appears blue.
(C) Lead (>50 ppb) ® It damage kidney and RBC.
(D) Fluoride (>2ppm) ® It causes brown mottling of teeth.
New answer posted
8 months agoNew answer posted
8 months agoWhich oxoacid of phosphorous has the highest number of oxygen atoms present in its chemical formula?
Contributor-Level 10
Pyrophosphorous acid = H4P2O5
Hypophosphorous acid = H4P2O6
Phosphoric acid = H3PO4
Pyrophosphoric acid = H4P2O7
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers