Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
Acidic oxide => Cl2O7
Neutral oxide => N2O, NO
Basic oxide => Na2O
Amphoteric oxide => As2O3
New answer posted
8 months agoContributor-Level 10
Emulsions gets separated into two layers on standing.
For stabilization of emulsion. Emulsifying agents added into it but not electrolyte.
New answer posted
8 months agoContributor-Level 10
Eq. a(1 - α) aα (aα/2)
Moles moles moles
Total no. of moles at equilibrium
= nA + nB + nC
New answer posted
8 months agoContributor-Level 10
BrF5 -> Br has sp3d2 hybridization
PCl5 -> P has sp3d hybridization
[Co (NH3)6]3+ -> Co has d2sp3 hybridization.
New answer posted
8 months agoContributor-Level 10
Degenerate orbitals must have same value of energy
Orbitals with same n and
New answer posted
8 months agoContributor-Level 10
Mass of C15H30 = volume * Density
= 1000 * 0.756 gm/
C15H30 + 22.5 O2 15CO2 + 15H2O
No. of moles of C15H30 = moles
No. of moles of O2 required =
Mass of O2 required = 22.5 *
No. of moles of CO2 liberated = 15 * moles
Mass of O2 liberated =
New answer posted
8 months agoContributor-Level 10
n-factor of KMnO4 in acidic medium = 5
n-factor of Mohr's salt = 1
meq of KMnO4 = meq of Mohr's salt
0.01 * 5 * V = 0.05 * 1 * 20
Volume of KMnO4 used, V = 20 mL
So; Volume of KMnO4 left in burette = 50 -20mL
= 30 mL
New answer posted
8 months agoContributor-Level 10
Moles of C = moles of CO2
=
Mass of C =
= 0.261g
Moles of H = 2 *
Mass of H =
Total mass of compound = 0.492g (given)
So; mass of O = (0.492 – 0.216 – 0.049) g
= 0.227g
% of O =
= 46.14%
the nearest integer = 46
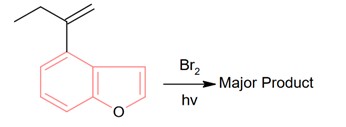
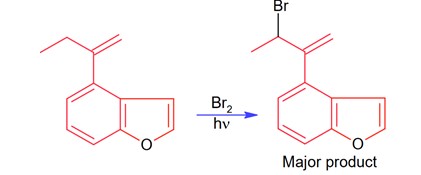
New answer posted
8 months agoContributor-Level 10
Bromination through free radical mechanism occurs at allylic carbon.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers