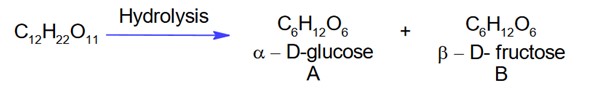Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
(a) will show cis- trans isomerism as;
(b) will not show cis- trans isomerism.
(c) will not show cis- trans isomerism.
(d) will not show cis-trans isomerism.
New answer posted
8 months agoContributor-Level 10
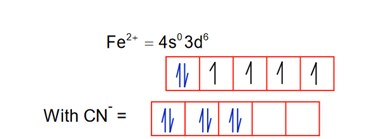
CN is a strong field ligand, so pairing occurs.
1.
So; it is diamagnetic
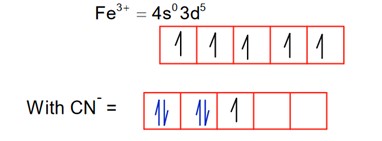
2.
So; it is paramagnetic.
3.
Ti3+ = 4s03d1
It is paramagnetic
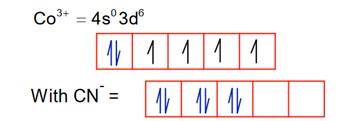
4.
It is diamagnetic
5.
Hence, are paramagnetic.
New answer posted
8 months agoContributor-Level 10
Molar mass of protein, M = 24751 g/mol
Molar mass of glycine = 75 g/mol
So; number of glycine units =
New answer posted
8 months agoContributor-Level 10
For combustion of Mg:
Mg (s) +
Here,
Now using
-601.7 =
So; magnitude of is 600 kJ (the nearest integer).
New answer posted
8 months agoContributor-Level 10
Using : PV = nRT
1.5 * 416 = n * 0.083 * 300
n = 25mol =
25 =
So, molar mass, M = 4 g/mol.
New answer posted
8 months agoContributor-Level 10
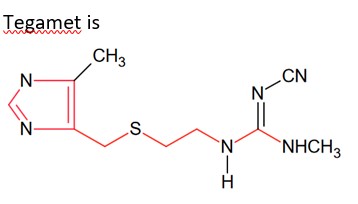
Compound X is sucrose as,
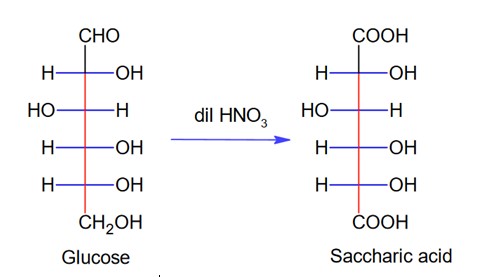
Here, glucose is A which on oxidation with HNO3 gives saccharic acid as
B is b-D fructose which is laevarotatory.
New answer posted
8 months agoContributor-Level 10
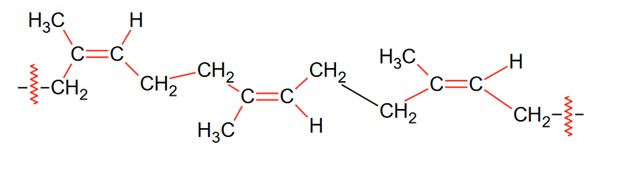
Natural rubber is a linear polymer of isoprene called cis-polyisoprene and has elastic properties.
Ci-polyisoprene molecule consists of various chains held together by weak vander Waals interactions and has a coiled structure.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers