Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
Mass of solute ; wB = 2.5 * 10-3 kg
Mass of solvent,
Boiling point of solution ;
Boiling point of water ;
So, elevation in boiling point,
Using ;
Molar mass of solute ; MB = 45 g/mol
New answer posted
8 months agoContributor-Level 10
Number of moles, n = 5 mol
Temperature, T= 300 K
Initial volume, V1 = 10L
Final volume, V2 = 20 L
Using;
Work done; w = -2.303 nRT log10
= -8630 J
So, magnitude of work done is 8630 J.
New answer posted
8 months agoContributor-Level 10
n = 1, 2, 3 …….;
A. n = 3
is incorrect as
B. n = 3 = -2 is correct set.
C. n = 2
= +2 is incorrect set as
So; correct set of quantum numbers is B and C.
New answer posted
8 months agoContributor-Level 10
Determining empirical formula of the given compound.
Mass Moles Simplest ratio
C 41.8g &n
New answer posted
8 months agoContributor-Level 10
A.
CO2 is colourless gas with brisk effervescence and turns lime water milky
B.
H2S + (CH3COO)2Pb →
C.
SO3 is colourless gas which turns acidified potassium dichromate solution green as
D. &nbs
New answer posted
8 months agoContributor-Level 10
A. Antipyretic reduces fever.
B. Analgesic reduces pain.
C. Tranquilizer reduces stress.
D. Antacid reduces acidity of stomach.
New answer posted
8 months agoContributor-Level 10
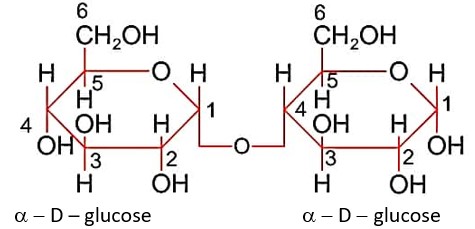
In maltose, two units of a-D-glucose are linked by glycosidic linkage at C1 and C4 as shown.
a - D – glucose a - D - glucose
Maltose has hemiacetal link so it is reducing sugar.
New answer posted
8 months agoContributor-Level 10
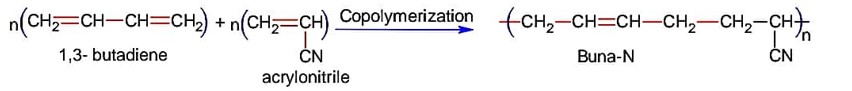
Buna-N is obtained by copolymerization of 1, 3-butadiene and acrylonitrile,
New answer posted
8 months agoContributor-Level 10
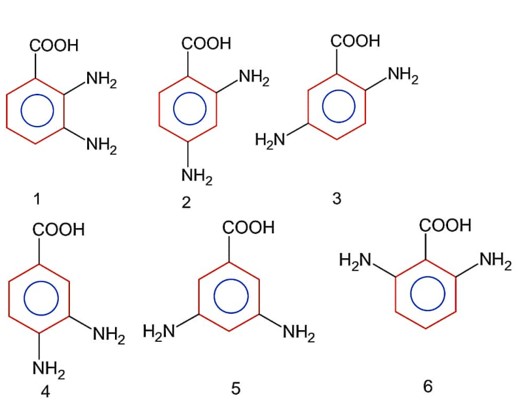
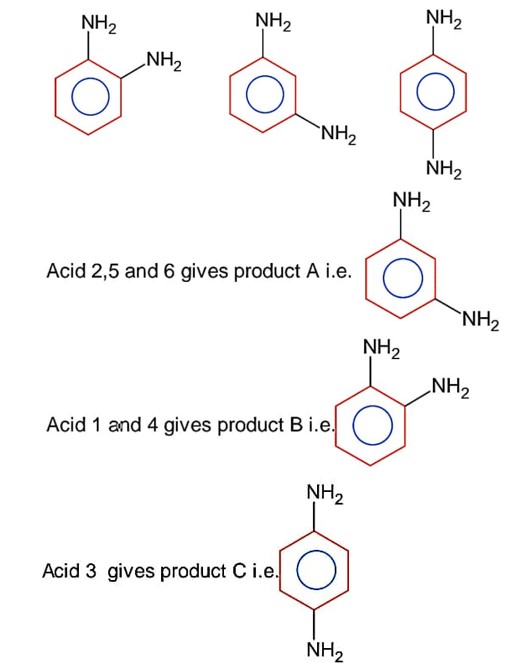
Above are the six possible forms of diaminobenzoic acid.
On decarboxylation we get;
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers