Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
6 months agoContributor-Level 10
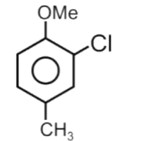
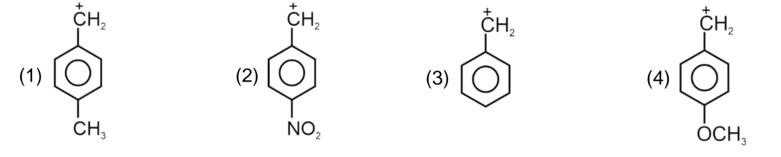
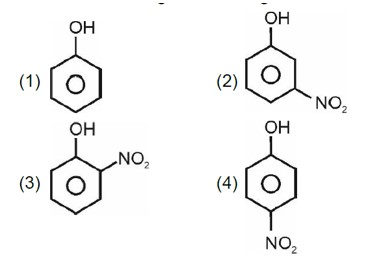
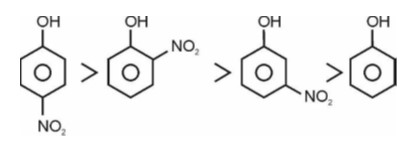
Electron withdrawing group decreases the stability of carbocation.
Correct order of stability of carbocation is
New answer posted
6 months agoContributor-Level 10
New answer posted
6 months agoContributor-Level 10
t1/2 = [A]1-n
for second order (n = 2) reaction : t1/2 is proportional to 1/ [A ]
New answer posted
6 months agoContributor-Level 10
More the number of ions in solution, more will be the specific conductance
Ionic conductivity of H+ is more than that of Na+
New answer posted
6 months agoContributor-Level 10
Li+ has the largest size in aqueous solution due to its high polarizing power.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers