Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
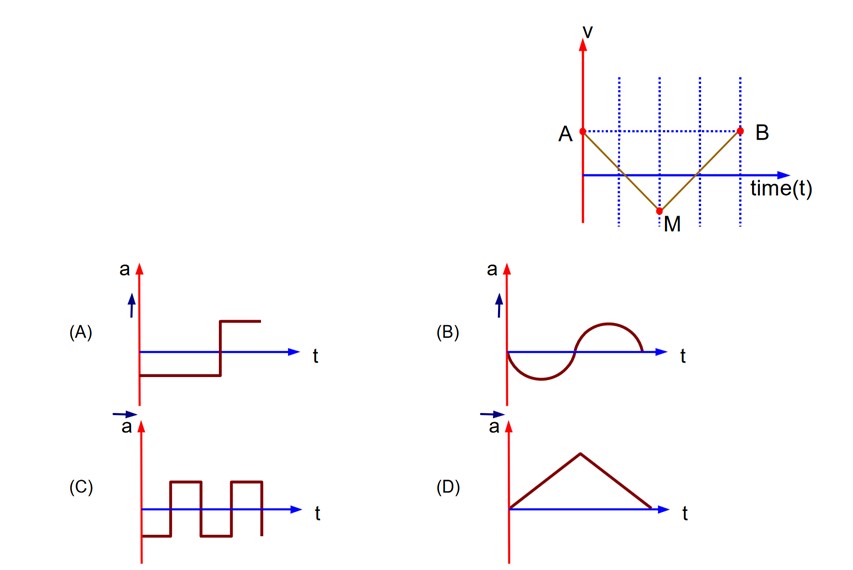
7 months agoContributor-Level 10
For part AM, slope of v – t graph is constant but negative. For part MB, slope of v – t graph is constant but positive.
New answer posted
7 months agoContributor-Level 10
For a reaction to be spontaneous;
So, minimum T at which reaction will be spontaneous is 200 K.
New answer posted
7 months agoNew answer posted
7 months agoContributor-Level 10
Initial1 mole -
At equilirbium 1-x mole x mole2x mole
V = 25 L and
T = 300 K.
At equilibrium, P = 1.9 atm
Total moles at equilibrium, n = 1 + 2x
V = 25 L
T = 300 K
Using, PV = nRT
1.9 * 25 = (1 + 2x) * 0.08206 * 300
x = 0.465
Now;Partial pressure of AB2 at equilibrium =
Using ;
= 0.728
KP = 0.73
Kp = 73 * 10-2
So; x = 73
New answer posted
7 months agoContributor-Level 10
Heavy water (D2O) is used in organic reaction to know the kinetics of reaction
Heavy water D2O is obtained by repeated electrolysis of H2O.
Heavy water has b.pt 101.4°C and ordinary water has b.pt 100°C
(b.pt of D2O > b.pt of H2O)
D2O has more molecular mass than H2O, so greater degree of association and hence greater b.pt and viscosity.
New answer posted
7 months agoContributor-Level 10
Vitamin E is helpful in delaying blood clotting while vitamin K help blood clotting.
New answer posted
7 months agoContributor-Level 10
Not all dipole – dipole interactions are responsible for hydrogen bonding, so assertion A is false. F is most E. N atom and also hydrogen bonds in HF are symmetrical.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers