Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoNew answer posted
8 months agoBeginner-Level 5
Students should prepare complete syllabus when they have time to prepare. However, you can use the list of highweightage chapters in last minute revision for scoring well.
- The p-Block Elements: This chapter holds a high weightage of 8–10 marks. (in the latest syllabus this is deleted)
Aldehydes, Ketones, and Carboxylic Acids: This chapter contributes around 8–10 marks.
Biomolecules: This chapter accounts for around 8 marks.
Chemical Kinetics: This chapter holds a high weightage of 5-6 marks.
The d- and f-Block Elements: This chapter contributes around 5-6 marks.
Amines: This chapter contributes around 5-6 marks.
New answer posted
8 months agoContributor-Level 10
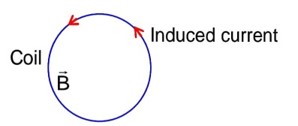
Using faraday's law magnetic field should be outward and decreasing with time
New answer posted
8 months agoContributor-Level 10
Name | Surname | College | Star |
Ram | Kumar | Chemical | 4 |
Komal | Sharma | Techno | 4 |
Rupali | Pandey | Anipal | 3 |
Kabeer | Gupta | Deccan | 4 |
Mansi | Verma | Barla | 5 |
New answer posted
8 months agoContributor-Level 10
Energy required to melt
Q =
->3.53 * 104 J
Heat produce in wire
H = l2RT
New answer posted
8 months agoBeginner-Level 5
Shiksha's NCERT notes are extremely useful for efficient preparation. We offer structured chapter-wise notes for the latest CBSE syllabus and provide concise summaries and key formulas. These notes are designed for quick and last-minute revision. These short revision notes offer step-by-step explanations for conceptual clarity and include important questions from previous years' papers and NCERT Textbooks. Our notes are equally beneficial for competitive exams like JEE Mains, NEET and other exams as well.
New answer posted
8 months agoContributor-Level 10
Room | Person |
101 | Krish, Ishan |
201 | Moksh/Nitin |
301 | Pari, Niti |
401 | Nitin/Moksh |
501 | Arjun |
601 | Jyoti |
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers