Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 9
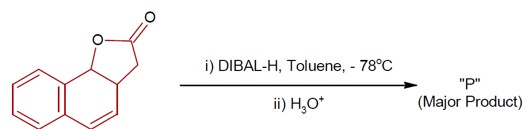
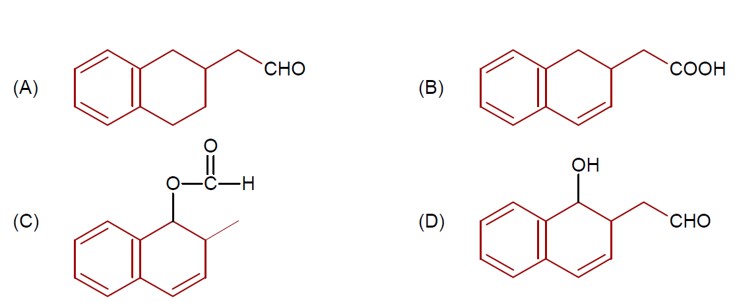
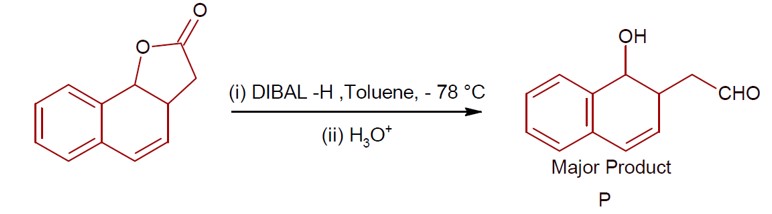
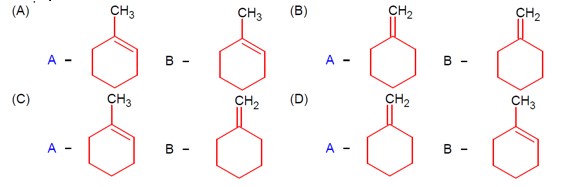
DIBAL-H at low temperature in a non-polar solvent, followed by hydrolysis, reduces esters to an aldehyde and an alcohol as a byproduct. The reaction shown is:
New answer posted
8 months agoContributor-Level 10
First, the number of unit cells in the given mass is determined. In an HCP structure, there are 6 atoms and 18 total voids (6 octahedral + 12 tetrahedral) per unit cell. Multiplying the number of unit cells by 18 gives the total number of voids. The result is14.9 x 10²¹, which is rounded.
New answer posted
8 months agoContributor-Level 10
Urea-formaldehyde resin is used for wood laminates because of its durable and unbreakable properties.
New answer posted
8 months agoContributor-Level 9
Solubility of CdSO? is water ; S = 8 * 10? M
Using Ksp = S²
Ksp = 64 * 10? M
Now,
CdSO? Cd²? + SO? ²?
in H? SO? (0.01M)
Ksp = [Cd²? ] [SO? ²? ]
64 * 10? = S? (S? + 0.01)
S? << 0.01
So, S? = 64 * 10? M.
New answer posted
8 months agoContributor-Level 9
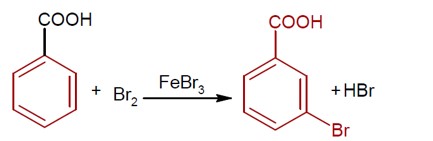
Moles of benzoic acid = 6.1 / 121 = 0.05
Theoretical moles of m- bromobenzoic acid = 0.05
Observed moles of m- bromobenzoic acid = 7.8 / 200 = 0.039
% yield = (0.039 / 0.05) * 100 = 78%
New answer posted
8 months agoContributor-Level 9
In Tollen's test for aldehyde, aldehyde is oxidized to carboxylic acid salt as:
R – CHO + H? O →R – COO? + 3H? + 2e?
So; 2e? are transferred per aldhyde group.
New answer posted
8 months agoContributor-Level 9
Partial hydrolysis of XeF? gives XeO? F?
XeF? + 2H? O → XeO? F? + 4HF
Compound A is XeF? , so number of lone pair on Xe is 1.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers