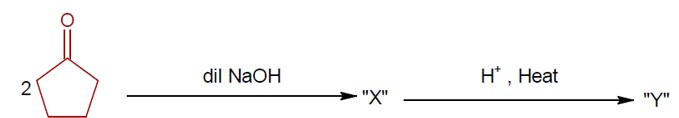
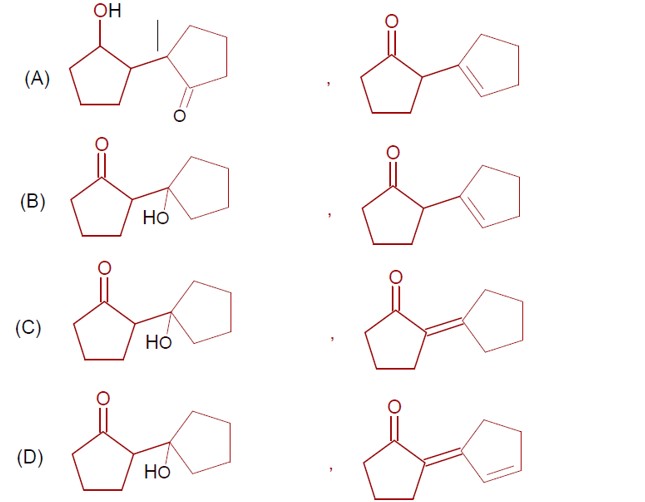
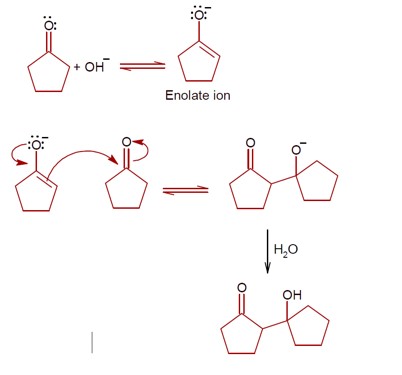
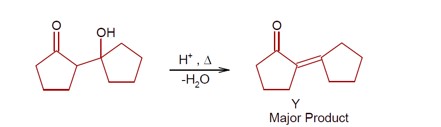
Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 9
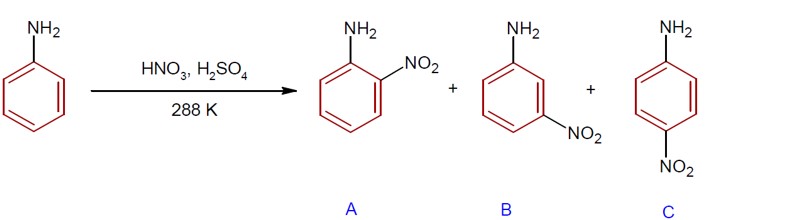
During nitration of aniline, meta- nitroaniline is also formed as product due to formation of –NH? group. The percentage of p, m and o product is 51%, 47% and 2% respectively
New answer posted
8 months agoContributor-Level 9
In Hoffmann bromamide reaction, hypobromite ion react with amide and in this reaction carbonyl group is lost as CO? ²? in form of Na? CO?
New answer posted
8 months agoContributor-Level 9
Match the following:
Antifertility drug → Norethindrone
Antibiotic → Salvarsan
Tranquilizer → Meprobamate
Artificial Sweetener → Alitame
New answer posted
8 months agoContributor-Level 9
Non- biodegradable wastes are generated by thermal power plants which produce fly ash. Bio-degradable detergents leads to eutrophication by decreasing oxygen level in water.
New answer posted
8 months agoContributor-Level 9
Covalent solids have high melting point due to strong covalent bonds, also they are insulator in both solid as well as in molten state.
New answer posted
8 months agoContributor-Level 9
In basic medium H? O? can behaves as both oxidizing and reducing agent, so it can oxidize Mn²? to Mn? and reduce I? to I?
New answer posted
8 months agoContributor-Level 9
Both C? H? OH and AgCN can generate nucleophile.
KCN generate nitrile as nucleophile while AgCN generate isonitrile as nucleophile in nucleophilic substitution reaction.
New answer posted
8 months agoContributor-Level 9
Given K_f = 1.85 K kg mol? ¹ for a solution with molality of 2 m.
ΔT_f = I * K_f * m
3.885 = I * 1.85 * 2
The van't Hoff factor, I = 1.05.
i = 1 + (n-1)α. For an electrolyte dissociating into 2 ions, n=2.
1.05 = 1 + (2-1)α.
The degree of dissociation, α = 0.05 or 50 * 10? ³.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers