Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
The reduction of aluminum oxide (Al? O? ) is performed through an electrolytic process in its molten state. This method is necessary because Al? O? is a highly ionic and stable compound.
New answer posted
8 months agoContributor-Level 9
m=2 molal
ΔT? = 100.52 – 100 = 0.52°C
Using, ΔT? = iK? m
0.52 = I * 0.52 * 2
i = 0.5
Now using, α = (1-i) / (1-1/n)
Where, n=2 (dimerisation)
α = (1-0.5) / (1-0.5) = 1
So, percentage association = 100%
New answer posted
8 months agoContributor-Level 10
In a C? fullerene molecule, all 60 carbon atoms are identical. Each carbon atom is bonded to three other carbon atoms via sigma bonds. The structure is composed of 20 six-membered rings and 12 five-membered rings.
New answer posted
8 months agoContributor-Level 10
In diacetamide (CH? CO)? NH), the lone pair of electrons on the nitrogen atom is delocalized through resonance with both adjacent carbonyl groups. This extensive resonance greatly decreases the electron density on the nitrogen atom.
New answer posted
8 months agoContributor-Level 9
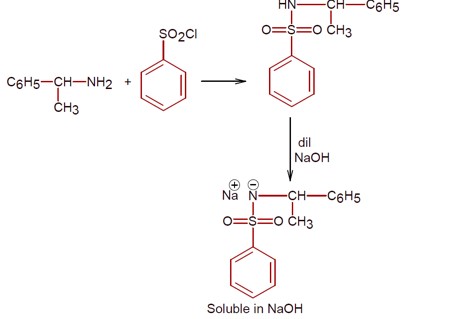
Only 1° amine react with benzene sulphonyl chloride to give a compound which is soluble in alkali
New answer posted
8 months agoContributor-Level 9
Metal sulphide sols are negatively charged while metal oxide sols are positively charged sols. So, CdS is negative while TiO? is positively charged sol.
New answer posted
8 months agoContributor-Level 10
Colloidal particles are small enough to pass through an ordinary filter but can be stopped by an ultrafilter paper due to their specific particle size range.
New answer posted
8 months agoContributor-Level 10
In the ammonolysis reaction, HCl is produced as a byproduct. To neutralize this acidic impurity, the mixture is treated with NaOH.
New answer posted
8 months agoContributor-Level 9
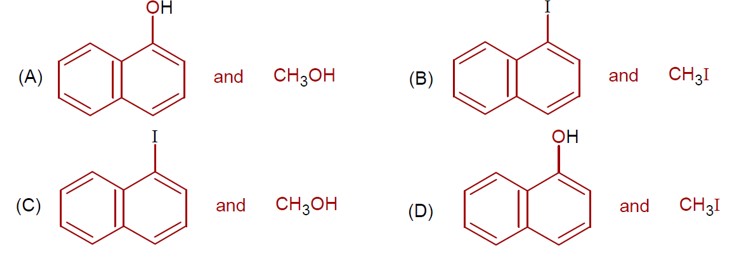
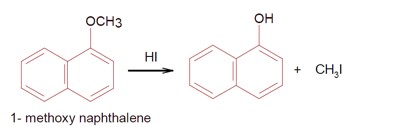
The following depicts a chemical reaction leading to a Major Product Y.
Mercury has low boiling point so is refined by distillation method.
Copper refining is done through electrolytic refining.
Silicon is refined by zone refining method.
Nickel is refined by vapour phase refining.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers