Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 9
A 6.5 molal solution means 6.5 moles of KOH is in 1 kg (1000 g) of solvent (H? O).
Moles of solute, n_B = 6.5
Mass of solute, W_B = 6.5 * 56 = 364 g
Mass of solvent, W_A = 1000 g
Mass of solution = 1364 g
Volume of solution = 1364 / 1.89 mL
Now, molarity = [6.5 / (1364 / 1.89)] * 1000 M = 9 M
New answer posted
8 months agoContributor-Level 9
Solubility product of A? X = 4S? ³
Where S? is the solubility of salt A? X.
Solubility product of MX = S? ²
Where S? is the solubility of MX.
Given 4S? ³ = 4 * 10? ¹² ⇒ S? = 10? M
Given S? ² = 4 * 10? ¹² ⇒ S? = 2 * 10? M
So, S? / S? = 10? / (2 * 10? ) = 50
New answer posted
8 months agoContributor-Level 9
Edge length in bcc, a? = 27 Å
Let, Edge length in fcc be a? Å
Now, the same element crystallises in bcc as well as fcc.
For bcc: 4r = √3 a? ⇒ r = (√3 / 4) a?
For fcc: 4r = √2 a? ⇒ r = a? / (2√2)
So, (√3 / 4) a? = a? / (2√2)
(√3 / 4) * 27 = a? / (2√2)
a? = 33.13 Å
The nearest integer is 33.
New answer posted
8 months agoContributor-Level 9
Roasting is a process in which sulphur is removed as SO? gas from sulphide ores on heating in excess of oxygen.
New answer posted
8 months agoContributor-Level 9
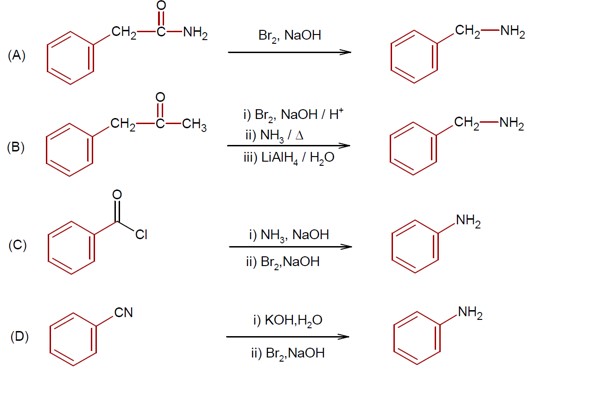
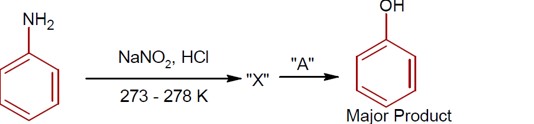
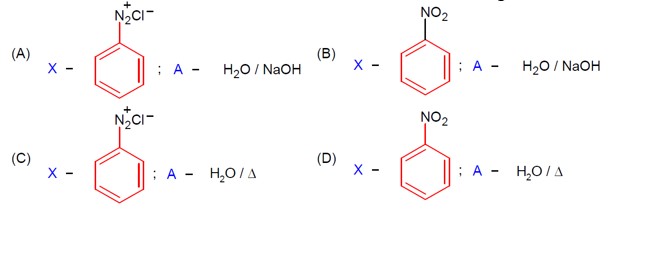
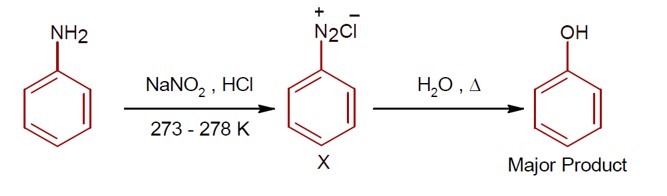
R-CONH? + Br? + 4NaOH → R-NH? + 2NaBr + Na? CO? + 2H? O
This reaction is the Hoffmann bromamide degradation, in which an amide is converted to a 1° amine.
New answer posted
8 months agoContributor-Level 9
The E° value for Ce? /Ce³? is +1.74 V, which suggests that Ce? is a strong oxidant, reverting to its common +3 oxidation state. So, Ce³? is more stable than Ce?
New answer posted
8 months agoContributor-Level 9
The size of the Bk³? ion is less than the Np³? ion because Berkelium (Bk) lies beyond Neptunium (Np) in the actinoid series, and the size variation here is because of the actinoid contraction.
New answer posted
8 months agoContributor-Level 9
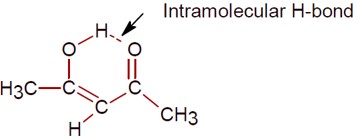
The enol form of acetone exists in less than 0.1% quantity, since its keto form is highly stable. But in the case of acetylacetone, the enol form is stabilized by intramolecular H-bonding, so its quantity increases to approximately 15%.
The intramolecular H-bond in the enol form of acetylacetone is shown.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers