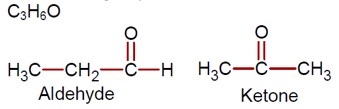
Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 9
When we add cryolite (Na? AlF? ) in the extraction of aluminium, the melting point of alumina decreases
New answer posted
8 months agoContributor-Level 10
Haematite: Fe? O?
Bauxite: Al? O? ·xH? O
Magnetite: Fe? O?
Malachite: CuCO? ·Cu (OH)?
New answer posted
8 months agoContributor-Level 9
Reaction Sequence:
o NO? + CH? COOH → HNO? + CH? COO?
o Sulphanilic acid (+ NH? CH? COO? ) + HNO? → N-acetylated diazonium intermediate + 2H? O
The intermediate + another molecule → Red Azodye + NH? + CH? COOH
New answer posted
8 months agoContributor-Level 10
100 mol of KBr is doped with 10? mol of SrBr?
Therefore, 1 mol of KBr contains 10? mol of SrBr?
Molar mass of KBr = 119 g/mol .
119 g of KBr contains 10? mol of SrBr?
1 g of KBr contains (10? / 119) mol of SrBr?
Each Sr²? ion introduced creates one cation vacancy to maintain electrical neutrality.
Number of cation vacancies = (moles of SrBr? ) * (Avogadro's number)
Number of vacancies = (10? / 119) * (6.023 * 10²³) = 5.06 * 10¹?
The answer provided in the document is "5 (Rounded off)", which likely refers to the coefficient 5.06.
New answer posted
8 months agoContributor-Level 10
For the dissociation of K? [Fe (CN)? ]? 4K? + [Fe (CN)? ]? , the number of ions produced (n) is 5.
The degree of dissociation (α) is related to the van't Hoff factor (i) by α = (i-1)/ (n-1).
Given α = 0.4: 0.4 = (i - 1) / (5 - 1) => 1.6 = I - 1 => I = 2.6
Using the depression in freezing point formula (ΔTf = i·Kf·m), and equating the ΔTf for two different solutions:
ΔTf (K? [Fe (CN)? ]) = ΔTf (A)
i? ·Kf·m? = i? ·Kf·m?
(2.6) * [ (18.1 / M) / (100-18.1)/1000] = (1) * [ (w? /M? ) / (W? /1000)]
The problem simplifies to finding the molar mass M of solute A:
2.6 * [18.1 / (M * 81.9)] * 1000 = [w? /M? ] * [1000/W? ]
Assuming the secon
New answer posted
8 months agoContributor-Level 10
3CaO + 2Al → 3Ca + Al? O?
ΔH? = ΣΔH? (Products) - ΣΔH? (Reactants)
ΔH? = [ (0) + (-1675) ] - [ (3 * -635) + (0) ]
ΔH? = -1675 - (-1905) = -1675 + 1905 = 230 kJ
Ans = 230
New answer posted
8 months agoContributor-Level 10
Cr? O? ²? + Fe²? - (H? )-> Cr³? + Fe³?
(n=6) (n=1)
Meq Cr? O? ²? = Meq Fe²?
20 * 0.03 * 6 = 15 * M * 1
M = (20 * 0.03 * 6) / 15 = 0.24 = 24 * 10? ²
Ans = 24
New answer posted
8 months agoContributor-Level 10
P? + 3NaOH + 3H? O → PH? + 3NaH? PO? (A)
Here, NaH? PO? is a reducing agent.
NaH? PO? + AgNO? + H? O → Ag + HNO? + NaH? PO?
Equivalents of NaH? PO? = equivalents of Ag (n-factor of NaH? PO? = 4)
1 mole * 4 = n moles of Ag * 1
So, moles of Ag = 4.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers