Physics
Get insights from 5.6k questions on Physics, answered by students, alumni, and experts. You may also ask and answer any question you like about Physics
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
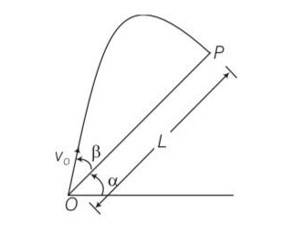
Explanation – particle is projected from the point O.
Let time taken in reaching from point O to point P is T.
for journey O to P
y=0,uy= Vosin ,ay= -gcos
y=uyt +
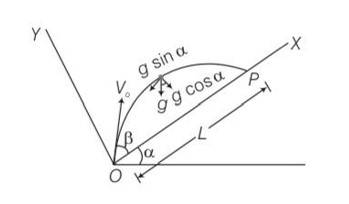
0= Vosin
T[Vosin T]=0
T = time of flight =
Motion along OX
x= L ,ux= Vocos , ax= -gsin
t =T =
x= uxt+
L= V0cos +
L= T[V0cos ]
L= [Vocos ]
L=
Z= sin
= sin
=
= ½ [sin2]
=
= [sin(2 )-sin ]
For z maximum
2 ,
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Volume occupied by 1 mole = 1mole of the gas at NTP= 22400mL=22400cc
So number of molecules in 1cc of hydrogen=
H2 is a diatomic gas, having a total of 5 degrees of freedom
So total degrees possesed by all the molecules
= 5
New answer posted
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Explanation – target T is at horizontal distance x= R+ and between point of projection y= -h
Maximum horizontal range R= …………1
Horizontal component of initial velocity = Vocos
Vertical component of initial velocity = -Vosin
So h = (-Vosin )t + 2………….2
R+ = Vocos
So t=
Substituting value of t in 2 we get
So h = (-V0sin )
H = -(R+ )tan +
, h = -(R+ )tan +
So h = -(R+ ) +
So h = -(R+ )+
So h = -R- +(R+ )
h=
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Number of helium = 5
T=7oC=7+273=280K
(a) hence number of atoms = number of moles Avogadro's number
= atoms
(b) now average kinetic energy per molecule = 3/2 KBT
= total internal energy
= number of atoms
=
New answer posted
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Explanation- speed of jackets = 125m/s
Height of hill = 500m
To cross the hill vertical component of velocity should be grater than this value uy=
So u2= ux2+uy2
Horizontal component of initial velocity ux =
Time taken to reach the top of hill t=
Time taken to reach the ground in 10 sec = 75 (10)= 750m
Distance through which the canon has to be moved =800-750=50m
Speed with which canon can move = 2m/s
Time taken canon = 50/2= 25s
Total time t= 25+10+10= 45s
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
When air is pumped, more molecules are pumped and boyle's law is staed for situation where number of molecules remains constant . in this case as the number of air molecules keep increases, hence mass change. Boyle's law is only applicable in situations, where number of gas molecule of remains fixed.
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Radius = 1Ao
Volume of hydrogen molecules = 4/3 r3
= 4/3 (3.14) (10-10)3 m3
Number of moles of H2 = mass/molecular mass=0.5/2=0.25
Molecules of H2 present = number of moles of H2 present
= 0.25
So volume of molecules present = molecule number volume of each molecules
= 0.25
6 3
PiVi= PfVf
Vf = i= 3
Vf= 2.7 3
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
(a) The average KE will be the same
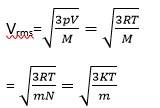
M= molar mass of the gas
m=mass of each molecular of the gas
R= gas constant
vrms
(b) k = Boltzmann constant
T= absolute temperature
mA>mB>mC
Vrms.A
New answer posted
10 months agoContributor-Level 10
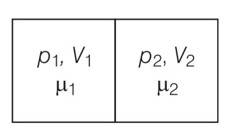
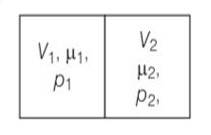
This is a short answer type question as classified in NCERT Exemplar
V1=2L ,V2=3L
µ1 = 4.0and µ2 =5.2
p1= 1.00 atm and p2 = 2.00 atm
p1V1= µ1RT1
p2V2= µ2RT2
when the partition is removed the gases get mixed without any loss of energy . the mixture now attains a common equilibrium pressure and total volume of the system is sum of the volume of individual chambers V1 and V2
, V =V1+V2
From the kinetic theory of gases pV=2/3 E
For mole 1 ,P1V1= 2/3
For mole 2 , P2V2= 2/3
Total energy is ( )= 3/2 ( )
PV==2/3Etotal = 2/3
P( )=
P= =
P=8/5 =1.6atm
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Mean free path l=1/
So n= number of molecules /volume
d = diameter of the molecule
l
d1=1Ao, d2=2Ao
l
l1:l2=4:1
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers