Physics
Get insights from 5.6k questions on Physics, answered by students, alumni, and experts. You may also ask and answer any question you like about Physics
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
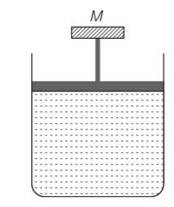
(c) The pressure inside the gas will be
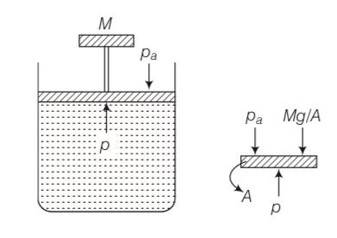
P= pa+Mg/A
A= area of piston
Pa= atmospheric pressure
Mg = weight of piston
When temperature is increases
pV=nRT so volume increases at constant pressure.
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(b) Boyle's law is applicable when temperature is constant
PV=nRT=constant
PV= constant
So pressure is inversely proportional to volume.
So process is called isothermal process.
New answer posted
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
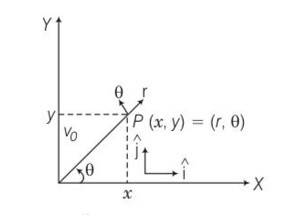
Explanation – r=cos ?…….1
…….2
Multiplying eq1 by sin and 2 with cos and adding
Rsin
= ?( )=j
= rsin
n(rcos )=i
b)r
= -cos
c)r=cos
dr/dt=d/dt(cos )=w[-cos ]
d)L= MoLT0
e)a=1unit , r=
v= dr/dt=
v=
= w
a=
a=
=
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
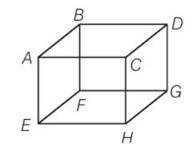
(d) In an ideal gas when a molecules collides elastically with a wall, the momentum transferred to each molecule will be twice the magnitude of its normal momentum. For the face EFGH, it transfer only half of that.
New answer posted
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Explanation- a) for x direction ux= u+vocos
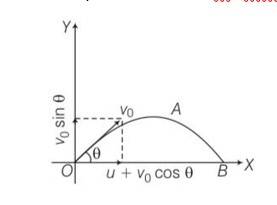
uy=velocity in y direction= v0sin
now tan
b) let t be the time flight y =0 uy=vosin
y= uyt+1/2 ayt2
0= vosin +
So T =
c) horizontal range R, = (u+vocos T= (u+vocos )
d) for range to be maximum dR/d
4vocos2
So cos =
e) cos =
so
f) if u=0 0
New answer posted
10 months agoContributor-Level 10
This is a multiple choice answer as classified in NCERT Exemplar
(b) As the motion of the vessel as a whole does not affect the relative motion of the gas molecules with respect to the walls of the vessel, hence pressure of the gas inside the vessel, as conserved by us, on the ground remains the same.
New answer posted
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
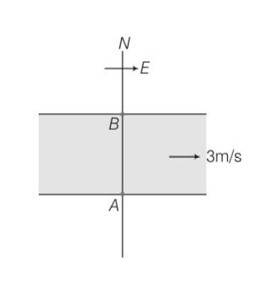
Explanation – speed of river Vr= 3m/s
Speed of swimmer Vs= 4m/s
(a) when swimmer starts swimming due north then its resultant velocity
V=
tan so 'N
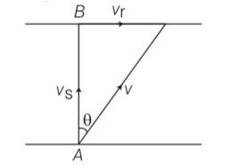
(b) to reach at point B resultant velocity will be
V=
tan
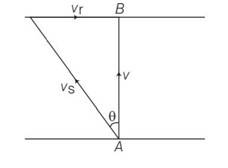
(c) time taken by swimmer t =d/v= d/4s
in case b time taken by swimmer to cross the river
t1=d/v=d/
so t
New answer posted
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Explanation – Vr= a? +b?
Velocity vg= 5m/s
Velocity of rain w.r.t girl = Vr-Vg= a? +b? -5?
= (a-5)? +b?
a-5=0, a=5
case II
vg = 10m/s?
Vrg= Vr - Vg
= a? +b? -10? = (a-10)? +b?
Rain appear to be fall at 45 degree so = b/a-10 =1
So b =-5
Velocity of rain = a? +b?
Vr = 5? -5?
Speed of rain Vr=
New answer posted
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
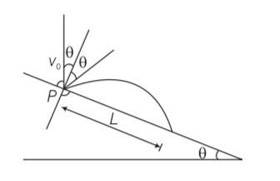
Explanation- y=O, uy= Vocos
ay=-gcos , t =T
applying equation of kinematics
y=uyt+ t2
0 = Vocos +T2
T=
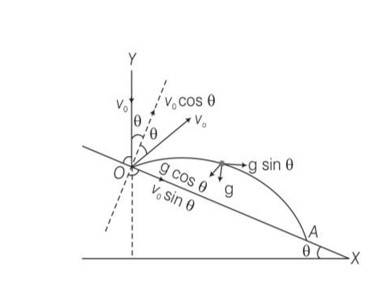
T= 2V0/g
X= L, ux=Vosin , ax= gsin , t=T=
X=uxt+
L= Vosin
L= sin
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
According to kinetic interpretation of temperature, absolute temperature of a given sample of a gas is proportional to total translational kinetic energy of its molecule.
Hence any change in absolute temperature of a gas will contribute to corresponding change in translational KE and vice versa.
N= number of moles
m=molar mass of the gas
when the container stops its total kinetic energy transferred to the gas molecules in the form of translational KE, thereby increasing the absolute temperature.
KE of molecules due to velocity KE=
Increase in translational KE =n T
Accordin
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers