Physics
Get insights from 5.6k questions on Physics, answered by students, alumni, and experts. You may also ask and answer any question you like about Physics
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Explanation- relaxation time =mean free path/rms velocity of electron
Also = = relaxation time is inversely proportional to velocities
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
For adiabatic change process we know
P1V1y= P2V2y
P (V+ )y = (P+ )Vy
PVy (1+ ) y=p (1+ )Vy
PVy (1+ ) PVy (1+ )
Y
dV=
hence work done increasing the pressure from P1 to P2
W=
=
W=
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Explanation – In the circuit when an electron approaches a junction, in addition to the uniform E that faces it normally (which keep the drift velocity fixed), as drift velocity (vd) is directly proportional to Electric field (E). That's why there are accumulation of charges on the surface of wires at the junction.
These produce additional electric fields. These fields alter the direction of momentum. Thus, the motion of a charge across junction is not momentum conserving
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Height of stairs h= 10m
Energy produced by burning 1 kg of fat = 7000Kcal
Energy produced by burning 5kg of fat = 5
Energy utilised in going up and down one time
= mgh + =
=
= 9000J= 9000/4.2=3000/1.4cal
Number of times, the person has to go up and down the stairs
= = 16.3 times
New answer posted
10 months agoContributor-Level 10
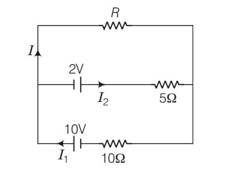
This is a Long Answer Type Questions as classified in NCERT Exemplar
Explanation- applying kirchhoff's junction rule I1 = I+I2
Applying kirchhoff's rule in outer loop containing 10V cell
10= IR+10I1……………. (1)
Applying kirchhoff's rule in outer loop containing 2V cell
2= 5I2-RI= 5 (I1-I)-RI
4= 10I1-10I-RI………… (2)
From 1 and 2
6=3RI+10I
2=I (R+10/3)
V= I (R+Reff)
After comparing V=2V, Reff= 10/3 ohm
Since effective internal resistance Reff of two cells 10/3 ohm, being the parallel combination 5 ohm and 10 ohm . the equivalent circuit is
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Temperature of the source T1= 500K and sink T2= 300K
Work done W= 1000J
Efficiency of Carnot engine = 1-T2/T1= 1-300/500= 200/500= 2/5
Efficiency = W/Q1
So Q1= W/efficiency = 1000
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
During driving temperature of the gas increases while volume remains constant. So according charle's law, at constant volume V.
Pressure is directly proportional to temperature. Therefore pressure of gas increases.
New answer posted
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Explanation- according to ohm's law V= IR
I= 6/6 = 1A
I= AneVd or Vd= i/neA
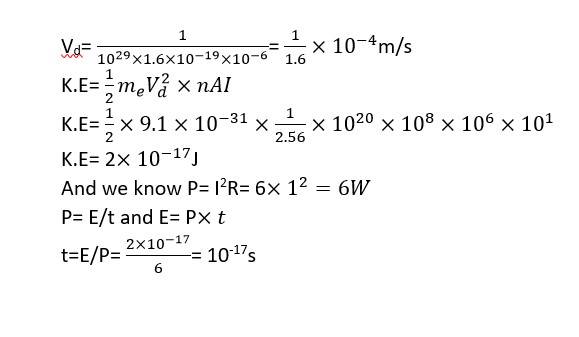
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Yes during adiabatic compression the temperature of a gas increases while no heat is
In adiabatic compression dQ=0
From the first law of thermodynamics dU= dQ-dW
dU=-dW
in compression work is done on the gas i.e work done is negative
dU=positive
hence internal energy of the gas increases due to which its temperature increases.
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
If a refrigerator's doors is kept open, then room will become hot, because amount of heat removed would be less than the amount of heat released in the room.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers