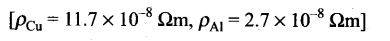Physics
Get insights from 5.6k questions on Physics, answered by students, alumni, and experts. You may also ask and answer any question you like about Physics
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
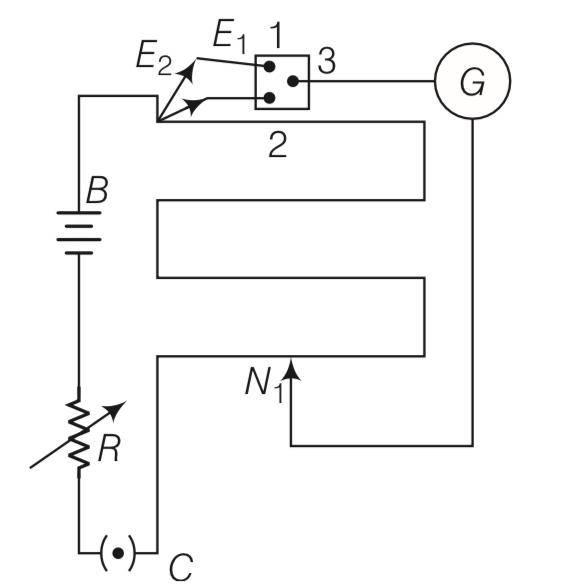
Explanation – let R' be the resistance of potentiometer wire.
Effective resistance of potentiomter and variable resistor r=50ohm is 500+R'
Effective voltage across potentiometer = 10V
The current through main circuit I= =
Potential difference across wire of potentiometer
IR'=
Since with 50 ohm resistor, null point is not obtained it is possible when
10R'
2R'<400 or R'<200 ohm
Similarly with 10 ohm resistor, null point is obtained its is only possible when
2R'>40
R'>40
7.5R'<80+8R'
R'>160
160
Any R' between 160 ohm and 200 ohm will achieve.
Since the null point on the last 4th segment of
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
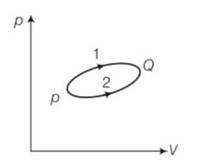
For path1
Heat Q1= 1000J
Work done =W1
For path 2
Work done W2= W1-100
As change in internal energy is same
dU=Q1-W1=Q2-W2
1000-W1=Q2-W1+100
Q2= 1000-100= 900J
New answer posted
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Explanation – power consumption in a day i.e in 5 = 10 units
Power consumption per hour = 2 units
Power consumption = 2 units =2KW= 2000J/s
Also power =V I
2000W= 220V l or l= 9A approx.
R=
Power consumption in first current carrying wire
P= I2R
l2= 1.7 10-8 j/s = 4J/s approx.
Loss due to joule heating in first wire = 100=0.2%
Power loss in Al wire =1.6 4= 6.4J/s
Fractional loss due to joule heating in second wire = 100= 0.32%
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Yes this is possible when the entire heat supplied to the system is utilised in expansion.
So its working against the surroundings.
New answer posted
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
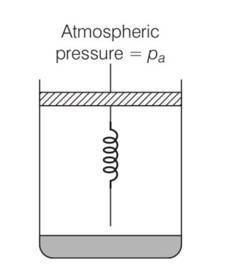
(a) Initially the piston is in equilibrium Pi=Pa
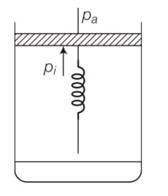
(b) On supplying heat , the gas expands from Vo to Vi
so increase in volume of the gas =Vi-Vo
as the piston is of unit cross sectional area hence extension in the spring
x=
force exerted by the spring on the piston= F= kx= K(Vi - Vo)
hence final pressure =Pf =Pa +kx
= Pa+K ( )
(c) From first law of thermodynamics
dQ=dU+dW
dU=Cv(T-To) = Cv(T-To)
T=
Work done by the gas =pdV+ increase in PE of the spring
= Pa(V1-Vo) + x2
dQ=dU+dW
= Cv(T-To)+Pa(V-Vo)+ x2
= Cv(T-To)+Pa(v-Vo)+1/2 ( )2
New answer posted
10 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Explanation – power consumption in a day i.e in 5 = 10 units
Power consumption per hour = 2 units
Power consumption = 2 units =2KW= 2000J/s
Also power =V I
2000W= 220V l or l= 9A approx.
R=
Power consumption in first current carrying wire
P= I2R
l2= 1.7 10-8 j/s = 4J/s approx.
Loss due to joule heating in first wire = 100=0.2%
Power loss in Al wire =1.6 4= 6.4J/s
Fractional loss due to joule heating in second wire = 100= 0.32%
New answer posted
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
Slope of the curve = f(V) , where V is the volume
Slope of P = f(V) curve at ((Po, V0 )= f(Vo)
Slope of adiabatic at (Po, V0 )= k(-Y)Vo-1-Y =-YPo/Vo
Now heat absorbed in the process P= f(V)
dQ=dU+dW= nCvdT+pdV
pV=nRT
T= pV/nR
T=
nCv
After solving we get
=
Heat is absorbed where dQ/dV>0 when gas expands
Hence YPo+Vof'(Vo)>0 or f'(Vo)>(-Y )
New answer posted
10 months agoContributor-Level 10
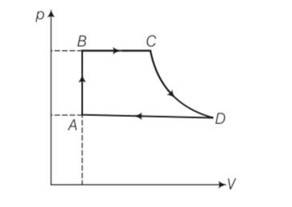
This is a long answer type question as classified in NCERT Exemplar
(a) For process AB
Volume is constant , hence work done dW=0
dQ=dU+dW=dU+0=dU
= nCvdT= nCv(TB-TA)
=
=
Heat exchanged =
(b) For process BC , p =constant
dQ= dU+dW =
heat exchanged =
(c) For process CD , because CD is adiabatic , dQ= heat exchanged =0
(d) DA involves compression of gas from VD to VA at constant pressure PA
heat transferred as similar way as BC1
hence dQ = PA(VA-VD)
New answer posted
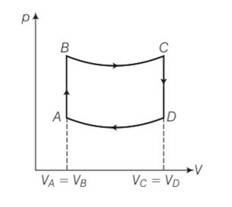
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
(a) For the process AB
dV=0 and dW=0
dQ=dU+dW=dU
dQ=dU= change in internal energy , so heat utilised is equal to change in internal energy.
Since p= in adiabatic temperature is directly proportional to pressure. So heat is supplied to the system in process AB.
(b) For the process CD volume is constant but the pressure decreases, hence temperature also decreases . so heat is also given to the surroundings.
(c) WAB= , WCD=
WBC=
= [pV]=
WDA=
B and C lies on adiabatic curve BC
PBVBY= PCVCY
PC = PB( )Y = PB( )Y= 2-YPB
Total work done by the engine in one cycle ABCD
New answer posted
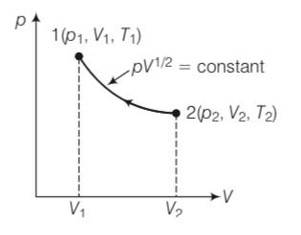
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
pV1/2= constant
P=k/
Work done from 1 to 2
W=
from ideal equation = pV=nRT
T= pV/nR=
T=
T1= , T1=
=
U=
= RT1( )
=2p1V11/2( )
= 2p1V11/2(2 )
= 2p1V1( )= 2RT1( )
=
= RT1( )+ 2RT1( )
=
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers