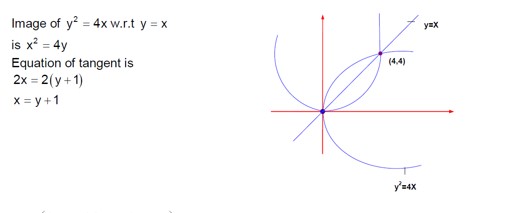
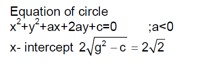Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 9
(|x| - 3)|x + 4| = 6

(-x - 3) (- (x + 4) = 6
(x + 3) (x + 4) = 6 ⇒ x² + 7x + 12 = 6 ⇒ x² + 7x + 6 = 0
(x + 1) (x + 6) = 0 ⇒ x = -6 (since x < -4)
Case (ii) -4 ≤ x < 0
(-x - 3) (x + 4) = 6
⇒ -x² - 7x - 12 = 6
⇒ x² + 7x + 18 = 0
The discriminant is D = 7² - 4 (1) (18) = 49 - 72 < 0, so no real solution.
Case (iii) x ≥ 0
(x - 3) (x + 4) = 6
⇒ x² + x - 12 = 6
⇒ x² + x - 18 = 0
x = [-1 ± √ (1² - 4 (1) (-18)] / 2 = [-1 ± √73] / 2
Since x ≥ 0 ⇒ x = (√73 - 1) / 2
Only two solutions.
New answer posted
8 months agoContributor-Level 9
Variance of a, b, c & a+2, b+2, c+2, are same.
Given: b = a + c (i)
d² = (1/3) (a² + b² + c²) - [ (a+b+c)/3]²
as a + c = b
d² = (1/3) (a² + b² + c²) - (2b/3)²
9d² = 3 (a² + b² + c²) - 4b²
⇒ b² = 3 (a² + c²) - 9d²
New answer posted
8 months agoContributor-Level 9
log? /? [ (|z|+11)/ (|z|-1)²] < 2
(|z|+11)/ (|z|-1)² > (1/2)²
(|z|+11)/ (|z|-1)² > 1/4
⇒ 4|z| + 44 > |z|² - 2|z| + 1
⇒ |z|² - 6|z| - 43 < 0
⇒ |z| - 7 ≤ 0
∴ |z|max = 7
New answer posted
8 months agoContributor-Level 10
The inequality is experience ( (|z|+3) (|z|-1) / (|z|+1) * log?2 ) ≥ log√? 16.
The right side is log? (1/2) 16 = log? (2? ¹) 2? = (4/-1)log?2 = -4. This seems incorrect.
Let's assume the base of the log on the right is √2. log√? 16 = log? (1/2) 2? = 2 * log?2? = 8.
The inequality becomes: 2^ (|z|+3) (|z|-1) / (|z|+1) ≥ 8 = 2³.
So, (|z|+3) (|z|-1) / (|z|+1) ≥ 3.
Let |z| = t. (t+3) (t-1) / (t+1) ≥ 3
t² + 2t - 3 ≥ 3t + 3
t² - t - 6 ≥ 0
(t-3) (t+2) ≥ 0
Since t = |z| ≥ 0, we must have t-3 ≥ 0.
So, t ≥ 3, which means |z| ≥ 3.
The minimum value of |z| is 3.
New answer posted
8 months agoContributor-Level 9
ΔG° = -9.478 kJ/mol
Using ΔG° = -2.303 RT log K_p
-9.478 * 10³ = -2.303 * 8.314 * 495 log K_p
1 = log K_p ⇒ K_p = 10
Here, for the given reaction A (g)? B (g), K_p = K_c
Initial A = 22 mmol
At equilibrium A = 22 - x mmol; B = x mmol
K_c = [B] / [A] = (x/V) / (22-x)/V) = x / (22-x) = 10
x = 10 (22-x) ⇒ x = 220 - 10x ⇒ 11x = 220 ⇒ x = 20
So, mmol of B at equilibrium are 20.
New answer posted
8 months agoContributor-Level 9
Moles of carbon in organic compound = Moles of carbon in CO?
n_c = 420 / 44 moles
Mass of carbon in organic compound = (420 / 44) * 12 = 114.54 g
Moles of hydrogen in compound = 2 * moles of H? O
n_H = 2 * (210 / 18) moles
Mass of hydrogen = 2 * (210 / 18) g = 23.33 g
% of H = (23.33 / 750) * 100 = 3.11%
The nearest integer is 3.
New answer posted
8 months agoContributor-Level 9
The balanced equation is:
2MnO? + 5C? O? ²? + 16H? → 2Mn²? + 10CO? + 8H? O
So, the value of c (coefficient for H? ) = 16.
New answer posted
8 months agoContributor-Level 9
H? O? can act as both an oxidizing & reducing agent in both acidic & basic medium. In the hydrogen economy, energy is stored & transmitted in the form of dihydrogen.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 690k Reviews
- 1850k Answers