Ncert Solutions Chemistry Class 11th
Get insights from 2k questions on Ncert Solutions Chemistry Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
6 months agoContributor-Level 10
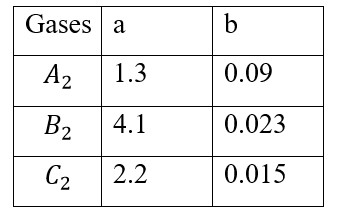
C? H? + 5O? → 3CO? + 4H? O
ΔH = − {6 (C = O) + 8 (OH)} + 2 (C – C) + 8 (C – H) + 5 (O = O)
= 8a? + 5a? + 2a? - 6a? - 8a?
New answer posted
6 months agoContributor-Level 10
(a) Li → used in bearings for motor engine.
(b) Na → used as coolant in fast breader nuclear reactor.
(c) K → used as absorbent of CO?
(d) Cs → used in photoelectric call.
New answer posted
6 months agoNew answer posted
6 months agoContributor-Level 10
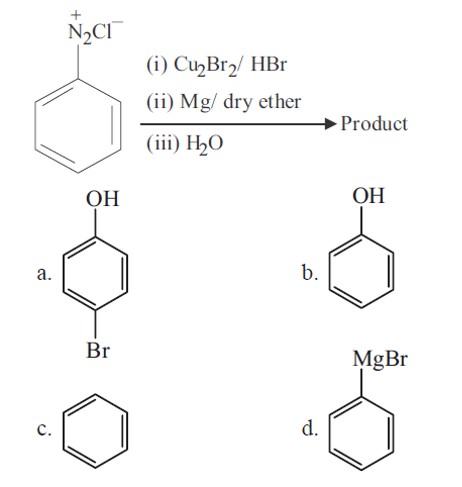
Bromobenzene is formed in first step (Sandmeyer reaction), which further gives phenyl magnesium bromide. Phenyl magnesium bromide further gives benzene with water.
+Mg (Br)OH
New answer posted
6 months agoContributor-Level 10
Helium is used as a diluent for oxygen in modern diving apparatus because of its very low solubility in blood.
New answer posted
6 months agoContributor-Level 10
In case, nitrogen and sulphur both are present in an organic compound, sodium thiocyanate is formed.
Na + C + N + S → NaSCN
Which further reacts as:
Fe³? + SCN? → [Fe (SCN)]²?
(Blood red color)
New answer posted
6 months agoContributor-Level 10
Coke- reducing agent
Diamond- sp³ carbons
Fullerenes- cage like structure
Graphite- used as lubricant
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers