Ncert Solutions Chemistry Class 11th
Get insights from 2k questions on Ncert Solutions Chemistry Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoContributor-Level 10
In sodium hydride (NaH), hydrogen has an oxidation state of -1. In this state, it can only act as a reducing agent.
New answer posted
7 months agoContributor-Level 9
Oxidation state of N
| NO | +2 |
|-|-|
| NO? | +4 |
| N? O | +1 |
| NO? | +5 |
So, order of oxidation state is
NO? > NO? > NO > N? O
New answer posted
7 months agoContributor-Level 9
Mn? O? is mixed oxide which contains MnO and Mn? O? , so Mn is in +2 and +3 oxidation state respectively. Both Mn? ² & Mn³? has unpaired electrons so Mn? O? will show magnetic property. While all other oxides have no unpaired electrons either on cation or on anion.
New answer posted
7 months agoContributor-Level 9
Be is used in X ray tube windows.
Mg is used in incendiary bombs & signals.
Compounds of Ca i.e CaCO? is used in extraction of metals like Fe.
Radium (Ra) is used in treatment of cancer in radiotherapy.
New answer posted
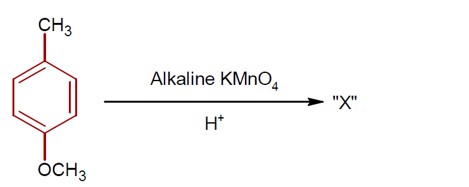
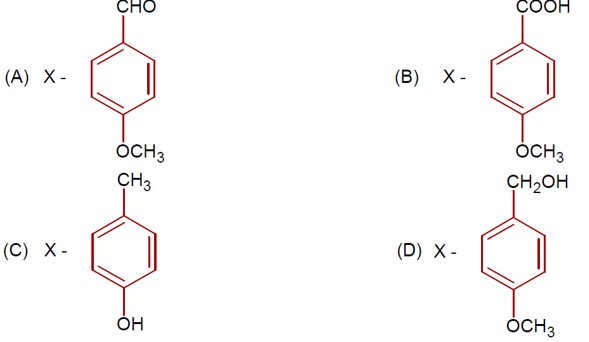
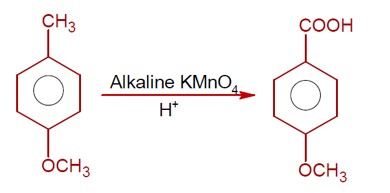
7 months agoContributor-Level 9
3-Hydroxy propanal
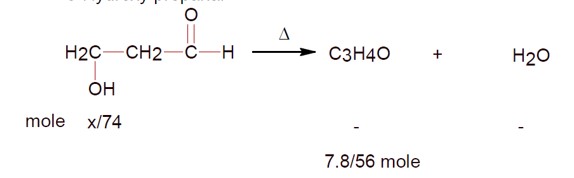
If 7.8g of C? H? O (molar mass 56 g/mol ) is formed, calculate the initial weight of 3-hydroxy propanal (molar mass 74 g/mol ).
Weight = (7.8/56) * 74 * (100/64) [Assuming 64% yield, though the number seems out of place].
Ans ≈ 16 g.
New answer posted
7 months agoContributor-Level 9
AX is a diatomic molecule with a bond order of 2.5.
The compound is NO. The total number of electrons = 15 (7+8).
New answer posted
7 months agoContributor-Level 10
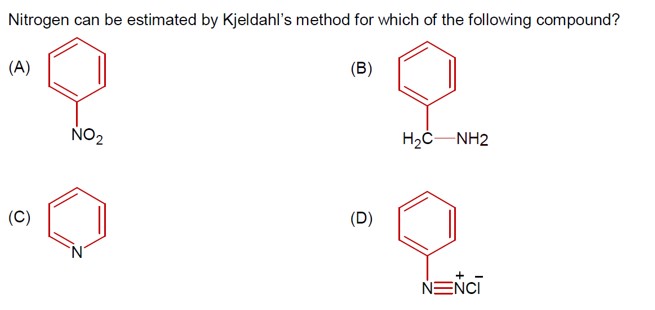
The Kjeldahl method is not applicable for nitrogen estimation in:
Compounds containing nitrogen in a nitro group.
Compounds containing an Azo group.
Pyridine.
New answer posted
7 months agoContributor-Level 10
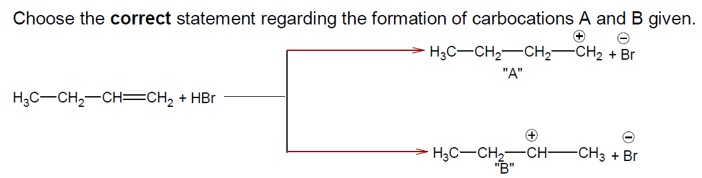
The addition of HBr to H? C−CH? −CH=CH? proceeds via carbocation formation.
Formation of a 1° Carbocation (A): H? C−CH?
Formation of a 2° Carbocation (B): H? C−CH? −C? H−CH?
The 2° carbocation (B) is more stable than the 1° carbocation (A). Therefore, the activation energy (Ea) for the formation of B is lower, and B is formed faster.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers