Ncert Solutions Chemistry Class 11th
Get insights from 2k questions on Ncert Solutions Chemistry Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoContributor-Level 9
o Alcoholic potassium hydroxide (Alc KOH):- used for β - elimination.
o Pd / BaSO? : - Lindlar's Catalyst.
New answer posted
7 months agoContributor-Level 10
A diagonal relationship is observed in the periodic table between elements of period (2) and period (3).
Li and Mg
Be and Al
B and Si
Li and Na do not show a diagonal relationship.
New answer posted
7 months agoContributor-Level 10
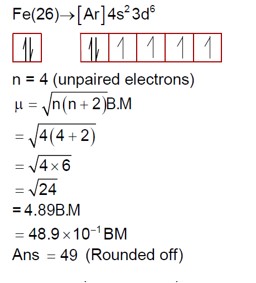
The electron configuration for Cr (24) is [Ar] 4s¹3d?
Chromium shows common oxidation states from +2 to +6.
New answer posted
7 months agoContributor-Level 10
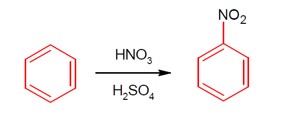
Moles of benzene = 3.9 g / 78 g/mol . This would produce (3.9 / 78) moles of nitrobenzene in 100% conversion.
Produced moles of nitrobenzene = 4.92 g / 123 g/mol .
% yield = [ (4.92 / 123) / (3.9 / 78) ] * 100 = [ (4.92 * 100 * 78) / (123 * 3.9) ] = 80.0%
Ans = 80
New answer posted
7 months agoContributor-Level 10
Molality = (mole of solute * 1000) / wt of solvent (gm)
100 = (n_solute * 1000) / [ (1 - n_solute) * 18]
(1 - n_solute) / n_solute = 1000 / (100 * 18) = 10/18
18 (1 - n_solute) = 10 n_solute
18 - 18 n_solute = 10 n_solute
18 = 28 n_solute
n_solute = 18 / 28? 0.6428 = 64.28 * 10? ²
Ans = 64 (Rounded off)
New answer posted
7 months agoContributor-Level 10
For the weak acid HA in the presence of strong acid HCl:
Ka = [ (Cα + 0.1) * Cα] / [C (1-α)] ≈ (0.1 * 10? ²α) / 10? ² = 0.1α
Given Ka = 2 * 10?
2 * 10? = 10? ¹ * α
α = 2 * 10?
Ans = 2
New answer posted
7 months agoContributor-Level 10
Mole of CH? = 6.4 / 16 = 0.4 and mole of CO? = 8.8 / 44 = 0.2
Total mole = (0.4 + 0.2) = 0.6 mole of a non-reacting mixture of gas
Using Ideal Gas Law; P = nRT / V
P = (0.6 * 8.314 * 300) / 10 = 149.65 kPa
Ans = 150 (Rounded off)
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers