Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
To determine the standard electrode potential of the system Mg2+|Mg, connect it to the standard hydrogen electrode (SHE). Keep the Mg2+|Mg system as cathode and SHE as cathode. This is represented as shown below.
Pt (s) | H2 (g, 1 bar)| H+ (aq, 1 M) |Mg2+ (aq, 1M)| Mg
The electrode potential of a cell is given by
E? = E? R – E? L
Where,
E? R- Potential of the half-cell in the right side of the above representation
E? L- Potential of the half-cell in the left side of the above representation
It is to be noted that the potential of the standard hydrogen electrode is zero.
Therefore, E? L = 0
E? = E? R – 0
⇒ E? = E? R
New answer posted
a year agoContributor-Level 10
4.3 The order of the reaction is sum of the powers of concentration of reactants in the rate law. According to this,
The order of the reaction = 1/2 + 2
= 2 1/2 or 2.5
The order of the reaction is 2.5
New answer posted
a year agoContributor-Level 10
4.2 Given-
Initial concentration (A1) = 0.5M
Final concentration (A2) = 0.4M
Time = 10 mins.
The formula for average rate of the reaction is,
rav = -1/2 X Δ{A} / Δt → Equation 1
? {A} = (A2)-( A1), the equation 1 is written as,
rav = -1/2 X A2 - A1 / Δt
= -1/2 X 0.4-0.5 / 10
= -1/2 X 0.1 / 10
= 0.005 mol L-1 min-1
= 5 * 10-3 mol L-1 min-1
The average rate of the reaction is 5 * 10-3 mol L-1 min-1
New answer posted
a year agoContributor-Level 10
4.1 Given-
Initial concentration (R1) = 0.03M
Final concentration (R2) = 0.02M
Time = 25 mins.
The formula for average rate of the reaction is,
rav = - Δ{R} / Δt → Equation 1
? {R} = (R2)-( R1), the equation 1 is written as,
rav = - R2 - R1 / Δt
= -0.02-0.03 / 25
= 4 X 10-4 mol L-1 min-1
The average rate of reaction in seconds is given by,
= 4 X 10-4 mol L-1 / 60 S
(dividing by 60 to convert minutes to seconds)
= 6.6 * 10-6 mol L-1 s-1
The average rate of the reaction in minutes is 4 * 10-4 mol L-1 min-1 and in seconds is 6.6 * 10-6 mol L-1 s-1
New question posted
a year agoNew answer posted
a year agoContributor-Level 10
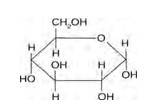
14.1
Glucose and sucrose are carbohydrates (optically active polyhydroxy aldehydes or ketones).
Structure of glucose:
Structure of sucrose:
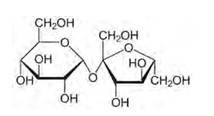
As you can see both the compounds have five –OH and eight –OH groups respectively. These –OH groups are responsible for the extensive hydrogen bonding with water. This –H bonding is responsible for the solubility of glucose and sucrose in water.
In case of cyclohexane or benzene (simple six-membered ring compounds), they do not contain any – OH groups. Hence, they cannot undergo –H bonding with water and are insoluble.
New answer posted
a year agoContributor-Level 10
15.24
Biodegradable polymers are designed to degrade into simpler components like water, CO2, Nitrogen etc. upon disposal by the action of living organisms. Extraordinary progress has been made in the development of practical processes and products from polymers such as starch, cellulose, and lactic acid. The need to create alternative biodegradable water- soluble polymers for down-the-drain products such as detergents and cosmetics has taken on increasing importance. Aliphatic polyesters are one of the important classes of biodegradable polymer. Example. Poly β-hydroxybutyrate – co-β-hydroxy valerate (PHBV)
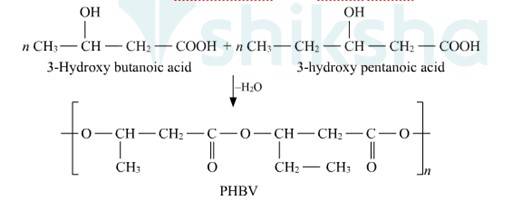
New answer posted
a year agoContributor-Level 10
15.23
Dacron is a polyester batting that should be added to any foam surface so that it will not be exposed directly to the fabric. Dacron has many indispensable qualities like batting reduces the friction foam has, and thus reduces wear to fabric and because polyester batting remains springy, it is ever ready to put some light pressure against fabric. This means that even as the fabric stretches with age (and always happens) batting will push against the fabric and keep wear-worn waves from developing.
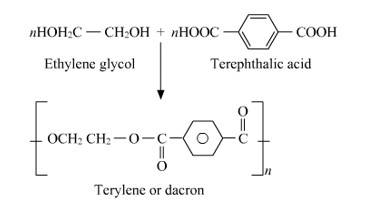
New question posted
a year agoNew question posted
a year agoTaking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers
