Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
(i) Ca2+ + 2e- → Ca
⇒ Here, 1 mole of Ca, i.e., 40g of Ca requires = 2 F electricity (F if Faraday)
∴ 20g of Ca requires = 20X2/40
= 1 F of electricity
Electricity in terms of Faraday required to produce 20.0 g of Ca from molten CaCl2 is 1 F of electricity.
(ii) Al3+ + 3e- → Al
⇒ 1 mole of Al, i.e., 27g of Al requires = 3 F electricity (F if Faraday)
∴ 40.0 g of Al will require = 3/27 X 40
= 4.44 F of electricity
Electricity in terms of Faraday required to produce 40.0 g of Al from molten Al2O3 is 4.44 F of electricit
New answer posted
a year agoContributor-Level 10
14.13
The condensation of the hydroxyl group of two monosaccharides to form a link between them is called glycosidic linkage. In other words, it refers to linkage developed between two different monosaccharide units through an oxygen atom by the loss of a water molecule. For example, in a sucrose molecule, two monosaccharide units, α-glucose and β–fructose, are joined together by a glycosidic linkage.
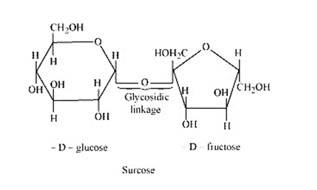

New answer posted
a year agoContributor-Level 10
The electrode reaction is given as,
Al3+ (aq) + 3e- → Al (s)
∴ The quantity of charge required for the reduction of 1 mol of Al3+ = 3F
= 3*96487 C
= 289461 C
The electrode reaction is given as,
Cu2+ (aq) + 2e- → Cu (s)
∴ The quantity of charge required for the reduction of 1 mol of Cu2+ = 2F
= 2*96487 C
= 192974 C
The electrode reaction is given as, MnO4→ Mn2+
i.e., Mn7+ + 5e - → Mn2+
∴ The quantity of charge required for the reduction of 1 mol of Mn7+ = 5F
= 5*96487 C
= 482435 C
New answer posted
a year agoContributor-Level 10
14.12
i. Ribose, 2-deoxyribose, galactose, and fructose are
Monosaccharides are the simplest units of carbohydrates which cannot be hydrolyzed into simpler compounds.
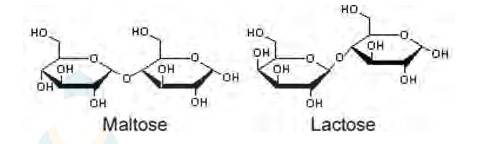
ii. Maltose and lactose are
A disaccharide is a carbohydrate that is formed when two monosaccharides are joined together and a molecule of water is removed from the structure. Lactose is a disaccharide formed from the combination of galactose and glucose.

New question posted
a year agoNew answer posted
a year agoContributor-Level 10
14.11
Carbohydrates are an essential compound for the organic life It is used as primary source of energy by plants and animals. It also fulfill other needs like synthesizing of other chemicals and provide the structure for cells within the body.
Energy is stored in it in the form of starch which, provide either complex or simple type of sugars. Complex sugars i.e., polysaccharides, give a constant supply of energy while simpler sugars, like monosaccharides, supplies a quicker jolt before dissolving.
Animals receive these starches through foods, especially those made from plant life such as grains and bread.
Plants manufacture their own ca
New answer posted
a year agoContributor-Level 10
Given -
Molarity, C = 0.00241 M
Conductivity, κ = 7.896 * 10–5 S cm–1
Molar conductivity? m =?
? 0m for acetic acid = 390.5 S cm2mol–1
Molar conductivity? m = k/c X 1000 S cm2 mol-1
= 7.896 X 10–5 S cm–1X 1000 cm3 L-1 / 0.00241 mol L-1
∴? m = 32.76 S cm2 mol-1
To calculate the dissociation constant, Ka, we use
Ka = → Equation 1
Here, we need to find the value of α (degree of dissociation), by the formula,

The molar conductivity? m is 32.76 S cm2 mol-1 and the dissociation constant, Ka is 1.86*10-5
New answer posted
a year agoContributor-Level 10
14.10
All those carbohydrates which reduce Fehling's solution to red precipitate of Cu2O or Tollen's reagent to metallic Ag is called reducing sugars. All monosaccharides (both aldoses and ketoses) and disaccharides except Sucrose are reducing sugars.
New answer posted
a year agoContributor-Level 10
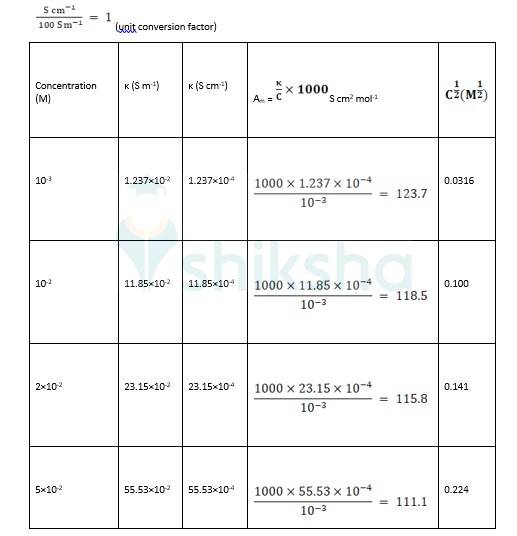
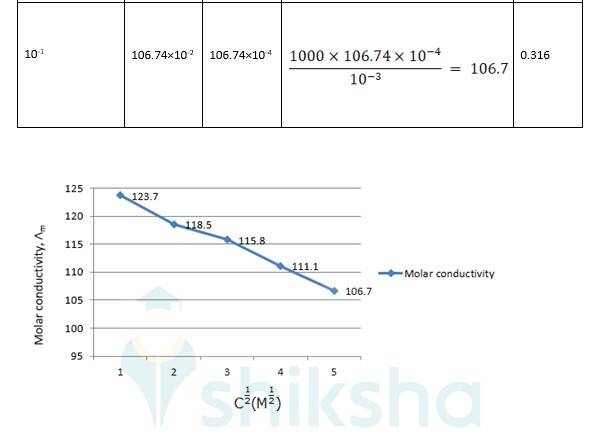
? 0 R = Intercept on the? axis = 124.0 S cm2 mol-1, which is obtained by extrapolation to zero concentration.
New answer posted
a year agoContributor-Level 10
14.9
Monosaccharides are the only the simplest units of carbohydrates and they are the simplest form of sugar. In other words, It is the most basic form of carbohydrates. They are made up of hydrogen, carbon and oxygen atoms. They are the building blocks of more complex carbohydrates i.e., they can join together and form complex carbohydrates.
For example:
i. Monosaccharides form
ii. 3-10 of them form
iii. 11 or more of them form
Monosaccharides are used to produce and store energy. Most organisms create energy by breaking down the monosaccharide glucose and harvesting the energy released from the bonds. Other Monosaccharides are
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers

