Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
14.4
Amino acids are organic compounds containing amine (basic) and carboxyl (acidic) functional group with a specific side chain. Both acidic and basic group are present in the same molecule. In, aqueous solution carboxyl group can lose a proton (H+) and amino group can accept a proton (H+) giving rise to the dipolar ion called as zwitter ion. Zwitter ion is shown below:
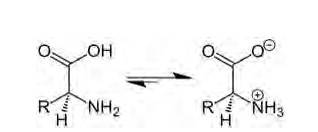
In this zwitter ion there is the presence of both positive as well as negative charge, so there is the development of strong electrostatic force of attraction between the molecules and the water. For this reason solubility of amino acids is higher. Due to strong e
New answer posted
a year agoContributor-Level 10
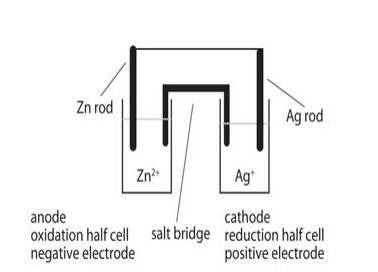
The galvanic cell corresponding to the given redox reaction can be represented as:
Zn|Zn2+ (aq)|Ag + (aq)|Ag
- 1) Zn electrode (anode) is negatively charged because, at this electrode, Zn is oxidized to Zn2+, causing electron accumulation at the
- 2) Electrons (ions) are the carriers of the current in the cell and in the external circuit, current flows from Ag (cathode) to Zn (anode) which is normally opposite to the electron flow which is from anode to cathode.
- 3) At anode:
Zn (s)⇒ Zn2 + (aq) + 2e– At cathode:
Ag + (aq) + e –⇒ Ag (s)
New answer posted
a year agoContributor-Level 10
14.3
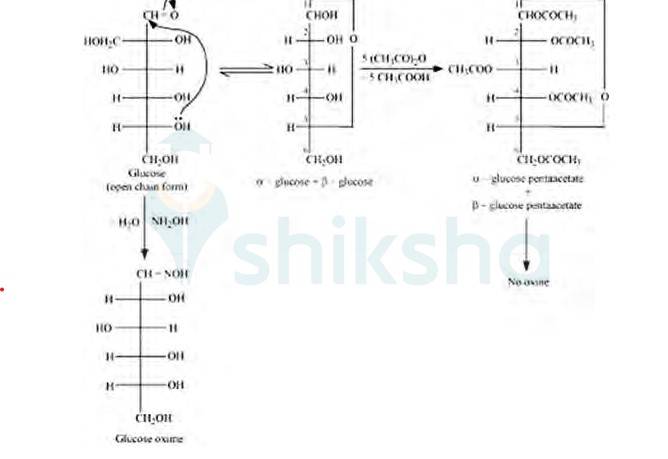
D-glucose reacts with hydroxylamine (NH2OH) to form oxime due to the presence of the aldehyde functional group (-CHO). This is due to the cyclic structure of glucose which forms an open chain structure in an aqueous medium, which then reacts to give an oxime.
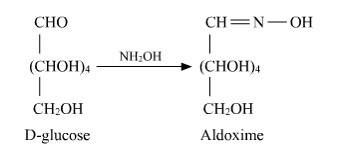
But in case of pentaacetate of D-glucose, it does not form open chain structure in an aqueous medium so it does not react with NH2OH.
New answer posted
a year agoContributor-Level 10
K + /K = –2.93V, Ag+ /Ag = 0.80V, Hg2+/Hg = 0.79V Mg2+/Mg = –2.37 V, Cr3+/Cr = – 0.74V
A 3.2 Reducing power of metals increase with the decrease of reduction potential. Hence, the increasing order of reducing power will be as,
Ag < Hg < Cr < Mg < K
When the reduction potential is lower, the element has more tendency to get oxidized and thus more will be reducing power. The metal that has more negative electrode potential will be the one with more reducing power. Thus, here potassium (K) has the highest reducing power among the given elements.
New answer posted
a year agoContributor-Level 10
The order in which the given metals displace each other from the solution of their salts is given by,
Mg>Al> Zn> Fe> Cu
A metal of stronger reducing power displaces another metal of weaker reducing power from its solution of salt. The order of increasing the reducing power of given metals is Cu< Fe< Zn< Algiven metals displace each other from the solution of their salts is given by, Mg>Al> Zn> Fe> Cu. This is hence arranged in decreasing order of its reactivity
New answer posted
a year agoContributor-Level 10
In the corrosion reaction, due to the presence of air and moisture, oxidation takes place at a particular point of an object made of iron. That spot behaves as the anode. The reaction at the anode is given by,
Fe (s) ⇒ Fe2+ (aq) + 2e-
Electrons released at the anodic spot move through the metal and go to another spot of the object, wherein presence of H+ ions, the electrons reduce oxygen. This spot behaves as the cathode. These H+ ions come either from H2CO3, which are formed due to the dissolution of carbon dioxide from the air into water. The cathodic reaction is given by
O2 (air) + 4Haq++4e-⇒ 2H O
The overall reaction is given by,
New answer posted
a year agoContributor-Level 10
Suggest two materials other than hydrogen that can be used as fuels in fuel cells.
New answer posted
a year agoContributor-Level 10
Anode: Lead (Pb)
Cathode: a grid of lead packed with lead oxide (PbO2)
Electrolyte: 38% solution of sulphuric acid (H2SO4)
The cell reactions are as follows :
Pb (s) + SO2-4 (aq) ⇒ PbSO4 (s) + 2e- (anode)
PbO2 (s) + SO2-4 (aq) + 4H+ (aq) +2e-⇒ PbSO4 (s) +2H2O (l) (cathode)
Pb (s) + PbO2 (s) +2H2SO4 (aq)⇒ 2PbSO4 (s) +2H2O (l)
(overall cell reaction)
On charging, all these reactions will be reversed.
New answer posted
a year agoContributor-Level 10
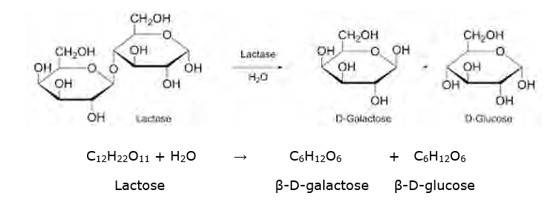
14.2
Lactose is a disaccharide carbohydrate (made up of two monosaccharide units) composed of β-D-galactose and β-D-glucose units. Hydrolysis breaks the glycosidic bond converting sucrose into β-D- galactose and β-D-glucose.
NOTE: But however, this reaction is so slow that it takes years for the solution of sucrose to undergo negligible change. Hence an enzyme called sucrase is added to proceed rapidly.
New answer posted
a year agoContributor-Level 10
Cr2O72– + 14H+ + 6e–⇒ 2Cr3+ + 7H2O
A 3.12
Cr2O72– + 14H+ + 6e–⇒ 2Cr3+ + 7H2O
For reducing one mole of Cr2O72–, 6 mole of electrons are required. Hence, 6 Faraday charges is needed. Hence, 6F = 6*96487 = 578922 C. Thus, the quantity of electricity is needed is 578922 C.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers
