Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
Metals with greater reactivity can be extracted electrolytically. Sodium, potassium, calcium, lithium, magnesium, aluminium which are present in the top of the reactivity series are extracted electrolytically.
New answer posted
a year agoContributor-Level 10
Current I = 0.5A
Time t = 2hrs = 2*60*60 = 7200 seconds
Charge Q = I * t
Q = 0.5*7200 = 3600 C
Charge carried by 1 mole of electrons (6.023*1023electrons) is equal to 96487C.
No of electrons = 6.023*1023 * 3600/96487
No of electrons = 2.25*1022 electrons
New answer posted
a year agoContributor-Level 10
C = 0.025 mol L-1
Am = 46.1 Scm2 mol L-1
λ0 (H+) = 349.6 Scm2 mol L-1
λ0 (HCOO-) = 54.6 Scm2 mol L-1
Λ0m (HCOOH) = Λ0 (H+) + Λ0 (HCOO-)
= 349.6 + 54.6
= 404.2 S cm2 mol L-1
Now, the degree of dissociation:

New question posted
a year agoNew answer posted
a year agoContributor-Level 10
The conductivity of a solution depends on the amount of ions present per volume of the solution. When diluted, the concentration of the ions decreases which implies that the number of ions per volume decreases thus, in turn, conductivity decreases.
New answer posted
a year agoContributor-Level 10
Given:
2Fe3+ (aq) + 2I- (aq) → 2Fe2+ (aq) + I2 (s)
E0cell = 0.236V
n = moles of e- from balanced redox reaction = 2
F = Faraday's constant = 96,485 C/mol
T = 298 K.
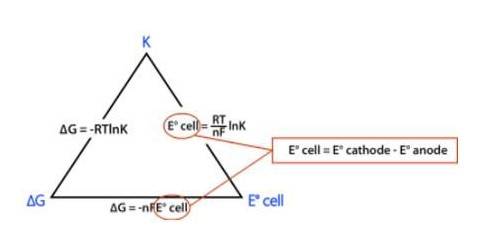
Using the formula, we get
? rG0 = – nFE0cell
⇒? rG0 = – 2 * FE0cell
⇒? rG0 = −2 * 96485 C mol-1 * 0.236 V
⇒? rG0 = −45540 J mol-1
⇒? rG0 = −45.54 kJ mol-1
Now,
? rG0 = −2.303RT log Kc
Where, K is the equilibrium constant of the reaction
R is the gas constant; R = 8.314 J-mol-C-1
⇒ −45540 J mol-1 = –2.303* (8.314 J-mol-C-1)* (298 K) * (log Kc)
Solving for Kc we get,
⇒ logKc = 7.98
Taking antilog both side, we get
⇒ Kc = Antilog (7.98)
⇒ Kc = 9.6 * 1
New answer posted
a year agoContributor-Level 10
Given:
[Ag+] = 0.002 M
[Ni2+] = 0.160 M
n = 2
(n = moles of e- from balanced redox reaction)
E0cell= 1.05 V
Now, using the Nernst equation, we get,

New answer posted
a year agoContributor-Level 10
Given:
For hydrogen electrode, pH = 10
n = 1
(n = moles of e- from balanced redox reaction)
On using the formula [H+] = 10– pH
⇒ [H+] = 10 − 10 M
We know,
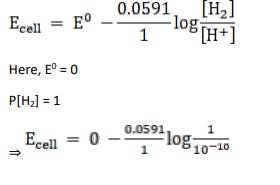
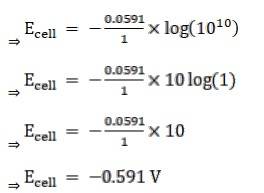
New answer posted
a year agoContributor-Level 10
For a substance to oxidise Fe2+ to Fe3+ ion, it must have high reduction potential than Fe3+. The reduction potential of Fe3+ to Fe2+ reaction is 0.77V, the substances which have reduction potentials higher than this value will oxidise Fe2+ ions. Comparing the values, from the table:

New answer posted
a year agoContributor-Level 10
NO, because Zn is very reactive with Cu. It reacts with copper sulphate to form zinc sulphate i.e., Zn displaces Cu and metallic Cu is also formed.
The reaction is given as:
Zn + CuSO4 ⇒ ZnSO4 + Cu
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers