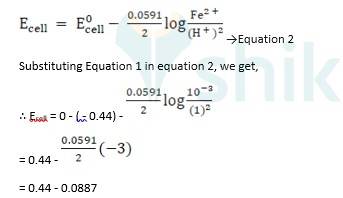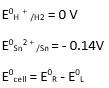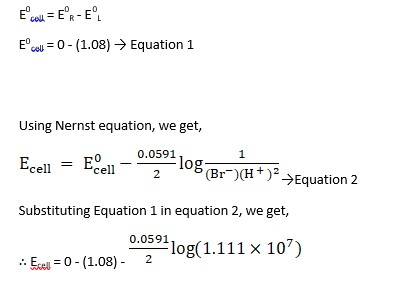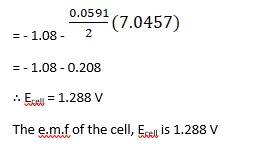Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
Given -
Resistance of a conductivity cell, R = 1500 Ω
Electrolytic conductivity of a solution, κ = 0.146 * 10-3 S cm-1
Cell constant =?
Conductivity, κ = cell constant/resistance
Cell constant = κ * R
= 0.146 * 10-3 S cm-1*1500 Ω
Cell constant = 0.219 cm-1
The cell constant of the cell containing 0.001M KCl solution at 298 K is 0.219 cm-1
New answer posted
a year agoContributor-Level 10
Given -
Molarity, C = 0.20 M
Electrolytic conductivity of a solution, κ = 0.0248 S cm-1
Molar conductivity =?
Molar conductivity, ∧m = K/C X 1000 S cm2 mol-1
=0.0248S cm-1 X 1000 Cm3L-1 / 0.20 mol L-1
∴ ∧m = 124 S cm2 mol-1
Molar conductivity (∧m) of 0.20 M solution of KCl at 298 K is 124 S cm2 mol-1
New answer posted
a year agoContributor-Level 10
14.8
A DNA is a double-stranded molecule (two polynucleotide chains whose nitrogenous bases are connected by hydrogen bonds).
In this molecule pairing or connection of bases occurs. Adenine always pairs with thymine, while cytosine always pairs with guanine.
So when DNA is hydrolyzed the amount of adenine produced is exactly equal to the amount of thymine produced, similarly, the amount of cytosine produced is equal to that of guanine.
But as given when RNA is hydrolyzed, there is no relationship between the quantities of different bases obtained. This suggests that RNA is a single-stranded molecule.
New answer posted
a year agoContributor-Level 10
The conductivity of a solution is defined as the conductance of one unit volume of solution kept between two platinum electrodes with a unit area of cross-section and at a distance of unit length.The molar conductivity of the solution is defined as the conducting power of all the ions produced by
one gram mole of an electrolyte in a solution. It is denoted by ∧m.
The conductivity of a solution (both for strong and weak electrolytes) always decreases with the decrease in concentration of the electrolyte i.e., on dilution. This pattern is seen because the number of ions per unit volume that carry the current in the solution d
New answer posted
a year agoContributor-Level 10
14.7
When a nucleotide from DNA containing thymine is hydrolyzed β-D-2 deoxyribose and phosphoric acid are obtained.
New answer posted
a year agoContributor-Level 10
Given - Zn → Zn2+ + 2e -, E0 = 0.76V (anode)
Ag2O + H2O + 2e - →2Ag + 2OH -, E0 = 0.344V (cathode), n = 2
ΔrG0 =?
Ecell0=?
Zn is oxidized and Ag2O is reduced.
Hence, the standard cell potential, Ecell0 is given as,
Ecell0 = ER0 - EL0
E0 cell = 0.344 + 0.76
∴ E0cell = 1.104 V
To calculate the standard Gibb's free energy? rG0, we use,
? rG0 = - nE0F → Equation 1
= - 2*96487*1.104 J
= - 213043.296 J
∴? rG0 = - 2.13*105 J
The standard cell potential, E0cell is 1.104 V and the standard Gibb's free energy? rG0 is - 2.13*105 J
New answer posted
a year agoContributor-Level 10
14.6
Vitamin C (ascorbic acid due to extensive –H bonding with water due to the presence of –OH group) is a water-soluble vitamin in contrast to vitamin A, D, E and K which are fat soluble. Also, humans cannot synthesize it due to the lack of specific enzyme and it is rapidly absorbed from the intestine.
Because it is water-soluble, it is not stored in our body to a significant amount but is readily excreted in the urine.
New answer posted
a year agoContributor-Level 10
(i) Mg(s)|Mg 2+(0.001M)Cu2+(0.0001 M)|Cu(s) (ii) Fe(s)|Fe 2+(0.001M)H+ (1M)|H2 (g)(1bar)| Pt(s) (iii) Sn(s)|Sn2+(0.050 M)H+ (0.020 M)|H2 (g) (1 bar)|Pt(s) (iv) Pt(s)|Br – (0.010 M)|Br2 (l )H+ (0.030 M)| H2 (g) (1 bar)|Pt(s).
A 3.5 Ecell = ?
(i) Mg + Cu2+ → Mg2+ + Cu (n = 2)
E0 Cu2+ / Cu+ = 0.34V
E0 Mg 2+ / Mg = - 2.37 V
Ecell0 = ER0-EL0
Ecell0 = 0.34 - ( - 2.37) → Equation 1
Using Nernst equation, we get,
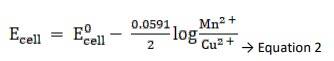
Substituting Equation 1 in equation 2, we get,
∴ Ecell = 0.34 - ( - 2.37) - 0.0591 / 2 log 10 -3/ 10-4
= 2.71 - 0.0591 / 2 log 10
= 2.71 - 0.02955
∴ Ecell = 2.68 V
The e.m.f of the cell, Ecell is 2.68 V
ii) Fe + 2H + →
New answer posted
a year agoContributor-Level 10
14.5
Eggs contain protein and proteins are a polymer in amino acids. When an egg is boiled the protein present inside the egg is denatured (the process of loss of biological activity of protein like their ability of –H bonding when subjected to a physical change like a change in temperature, pH) and coagulated (the process of liquid changing to a solid or semi-solid state). Due to this, the water present in the egg is absorbed by the coagulated protein by –H bonding.
New answer posted
a year agoContributor-Level 10
(1) Known - E0Cr3+/Cr = - 0.74 V
E0 cd2+ = - 0.40 V
? rG0 =? K =?
The galvanic cell of the given reaction is written as - Cr (s)|Cr3+ (aq)| Cd2+ (aq)|Cd (s)→ Reaction 1
Hence, the standard cell potential is given as, E0 = ER0 - EL0
= - 0.40 - (- 0.74)
∴ E0 = + 0.34 V
To calculate the standard Gibb's free energy? rG0, we use,
? rG0 = - nE0F → Equation 1
wherenF is the amount of charge passed and E0 is the standard reduction electrode potential. Substituting n = 6 (no. of e - involved in the reaction 1), F = 96487 C mol-1,
E0 = + 0.34 V in Equation 1, we get, l
? rG0 = - 6*0.34V*96487 C mol-1
= - 196833.48 CV mol-1
= - 196833.48 J mol-1
∴?
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers