Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
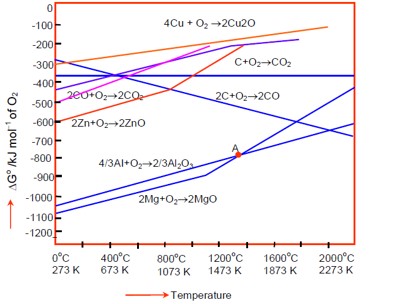
At the point of intersection in an Ellingham diagram? G for two processes becomes equal, so? G for the reduction becomes zero. A sudden increase in the slope indicates a change in the state of the metal oxide, i.e., from solid to liquid or liquid to vapor.
New answer posted
8 months agoContributor-Level 10
In the ammonolysis process, bond cleavage is carried out in the presence of NH? When a halide compound is treated with NH? , the halide ion (X? ) is substituted by an amino group (NH? ) in a nucleophilic substitution reaction.
New answer posted
8 months agoContributor-Level 10
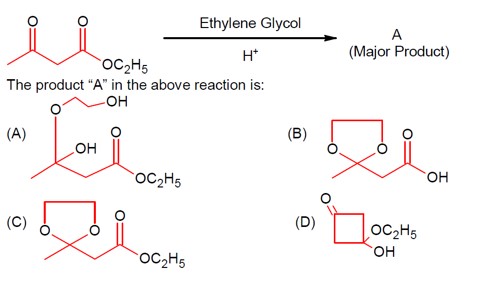
A more reactive carbonyl, i.e., a ketone, is masked with ethylene glycol first in an acidic medium.
New answer posted
8 months agoContributor-Level 10
Allosteric inhibitors are a type of drug-enzyme inhibitor that binds to a site other than the active site of the enzyme, causing a conformational change that affects the active site. The statement in the document incorrectly states they bind at the active site due to steric factors.
New answer posted
8 months agoContributor-Level 10
A divalent metal ion with an atomic number (Z) of 25 is Mn²?
Mn²? (Z=25) has the electronic configuration: 3d? 4s? (n=5, five unpaired electrons).
The magnetic moment (μ) is calculated as: μ = √ [n (n+2)] = √ [5 (5+2)] = √35 = 5.92 BM.
New answer posted
8 months agoContributor-Level 10
Solid sol is a colloidal system consisting of a gas dispersed in a solid, e.g., pumice stone, foam rubber.
New answer posted
8 months agoContributor-Level 10
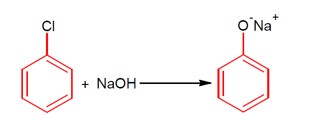
This describes a nucleophilic aromatic substitution reaction. Aryl halides are very less reactive toward this reaction, so the reaction takes place at a high temperature, i.e., 623K, and high pressure of 300 atm.
New answer posted
8 months agoContributor-Level 10
Ellingham diagram provides information on Gibb's free energy for formation of oxides as a function of temperature.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers