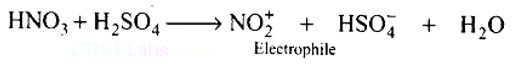Amines
Get insights from 162 questions on Amines, answered by students, alumni, and experts. You may also ask and answer any question you like about Amines
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
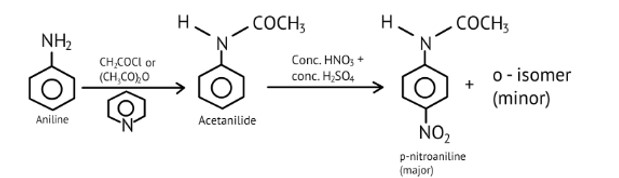
Ans: As −NH2 is a strong activating group, the aniline will readily undergo electrophilic substitution reaction, and it is difficult to cease reaction at the mono substitution stage.
Therefore, the activating group −NH2 is protected by an acetylation process.
The acetylated complex formed utilizes the lone pair of nitrogen and are less available for donation, this helps to carry out the nitration reaction easily.
New answer posted
10 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
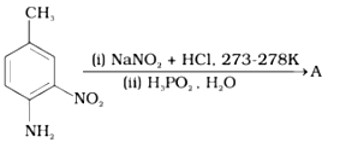
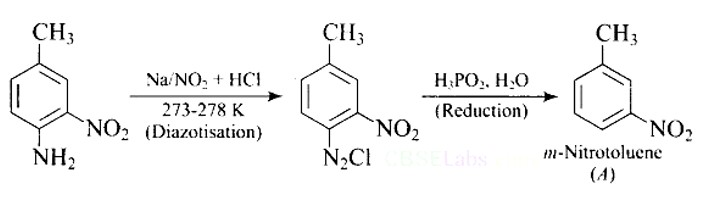
Ans: The reaction of aniline with nitrous acid at 273-278 K produces benzene diazonium chloride. The reaction of sodium nitrite with hydrochloric acid produces nitrous acid in the reaction mixture. Diazotisation is the process of converting primary aromatic amines into diazonium salts. Because of its instability, the diazonium salt is generally not stored or used immediately after preparation.
The crystalline solid benzene diazonium chloride is colorless.
It is easily soluble in water and stable at room temperature, but it reacts with water when warmed.
In the dry state, i
New answer posted
10 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans: Hinsberg's reagent is also known as benzenesulphonyl chloride (C6H5SO2Cl). When it reacts with primary and secondary amines, it produces sulphonamides. Allowing secondary and tertiary amines to react with Hinsberg's reagent allows them to be distinguished (benzenesulphonyl chloride C6H5SO2Cl). Secondary amines react with Hinsberg's reagent to form an alkali-insoluble product. N, N-diethylamine, for example, reacts with Hinsberg's reagent to form N, N-diethylbenzenesulphonamide, which is insoluble in alkalis. Tertiary amines, on the other hand, are unaffected by Hin
New answer posted
10 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans:
New answer posted
10 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans: The best reagents for the conversion of nitrile to primary amine are LiAlH4 and Sodium/Alcohol. By reduction, the nitriles can be converted into a corresponding primary amine.
New answer posted
10 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
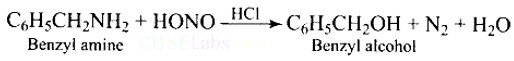
Ans: When Benzyl amine is treated with nitrous acid, firstly BDC is formed which is unstable and decomposes to Benzyl alcohol.
New answer posted
10 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans:
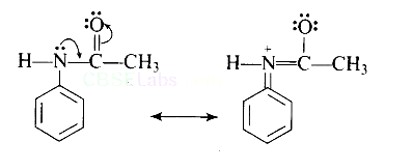
Direct nitration of aniline is not possible on account of oxidation of -NH2 group. However, nitration can be carried after protecting the -NH2 group by acetylation to give acetanilide which is then nitrated and finally hydrolysed to give o- and p-nitroanilines.
The acetyl group being electron withdrawing attracts the lone pair of electrons of the N-atom towards carbonyl group.
As a result, the activating effect -NH2 group is reduced i.e., the lone pair of electrons on nitrogen is less available for donation to benzene ring by resonance. Therefore, acti
New answer posted
10 months agoContributor-Level 10
This is a Short Type Questions as classified in NCERT Exemplar
Ans: HNO3 acts as a base in the nitrating mixture (HNO3 + H2SO4) and provides the electrophile.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Option (A and B)
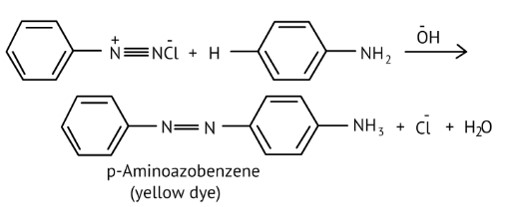
Benzene diazonium chloride reacts with phenol to form p-hydroxy azobenzene by coupling the phenol molecule at its para position with the diazonium salt. This is referred to as a coupling reaction. This is an illustration of an electrophilic substitution reaction.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: a and b
In addition to the nitro derivatives, direct nitration of aniline produces tarry oxidation products. Furthermore, aniline is protonated in the strongly acidic medium to form the anilinium ion, which is meta directing. As a result, in addition to the ortho and para derivatives, a significant amount of meta derivative is formed.
However, by protecting the? NH2 group with an acetylation reaction with acetic anhydride, the nitration reaction can be controlled and the p-nitro derivative obtained as the main product.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 690k Reviews
- 1850k Answers