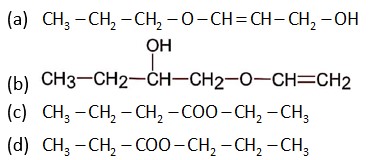Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoContributor-Level 10
Not all dipole – dipole interactions are responsible for hydrogen bonding, so assertion A is false. F is most E. N atom and also hydrogen bonds in HF are symmetrical.
New question posted
7 months agoNew answer posted
7 months agoContributor-Level 10
Hydrogen peroxide reduces iodine to iodide ion is basic medium as;
New answer posted
7 months agoContributor-Level 10
Here A is CH3 - CH2 – CH2 – COO – CH2 – CH3
The given sequence of reaction is:
New answer posted
7 months agoContributor-Level 10
Ce, Pr, Nd, Tb and Dy are the only lanthanoids which shows + 4 O.S, so can form MO2, but Yb does not shows +4 O.S so can't form MO2.
New answer posted
7 months agoContributor-Level 10
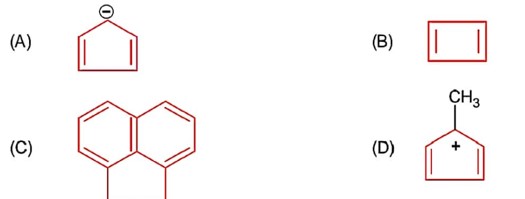
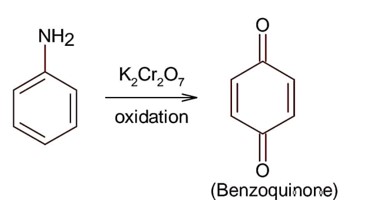
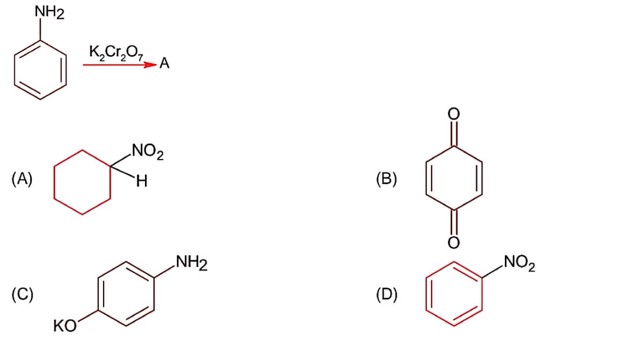
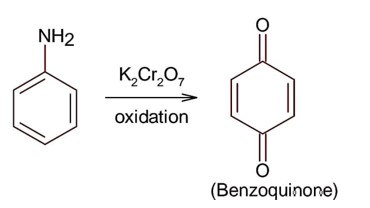
Only 1° aromatic amines give stable diazonium salt on reaction with nitrous acid
Here 1° aromatic amine is 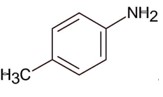
New answer posted
7 months agoContributor-Level 10
N > O > Be > B
Due to half filled configuration N has more I.E than oxygen and due to fully filled configuration Be has more I.E than B.
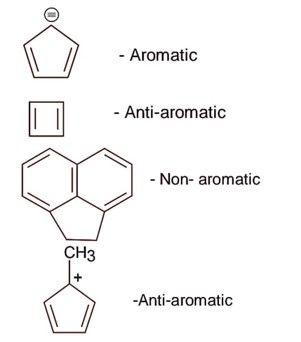
(a) → (ii) (b) → (iii) (c) → (iv) (d) → (i)
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers