Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New question posted
7 months agoNew answer posted
7 months agoContributor-Level 10
Nowdays liquid CO2 used as washing clothes, because Cl2 and C2H2Cl4 are toxic
New answer posted
7 months agoContributor-Level 10
An alkali metal oxide of formula MO2 is super oxide which is formed by K and Rb. KO2 has pale yellow colour and paramagnetic nature, while alkaline earth metal oxides formula MO2 are peroxide and diamagnetic nature.
New answer posted
7 months agoContributor-Level 10
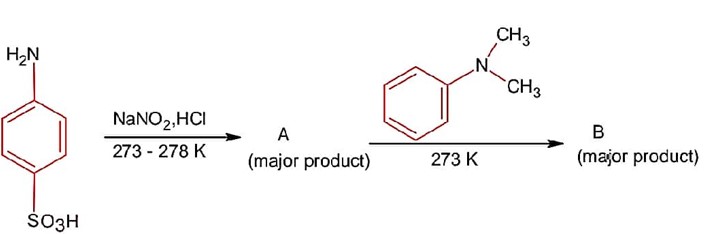
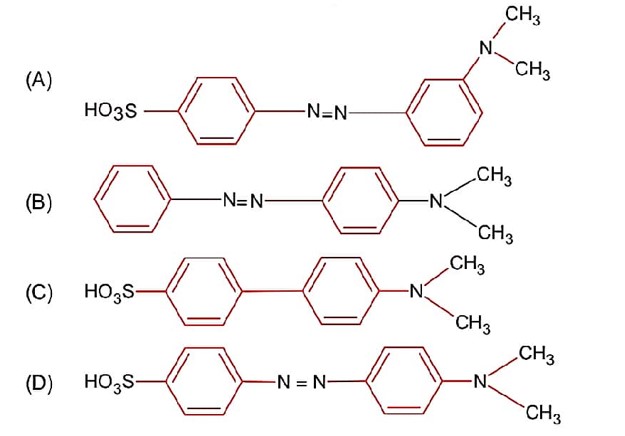
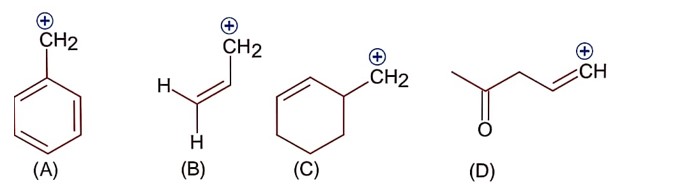
Only (A) and (B) structures have resonance effect. In (A) benzyllic resonance and in (B) allylic resonance of carbocation.
New answer posted
7 months agoContributor-Level 10
Enzymes are the form of proteins and highly specific regarding temperature and pH change. It also lowers the activation energy for biochemical reaction.
New answer posted
7 months agoContributor-Level 10
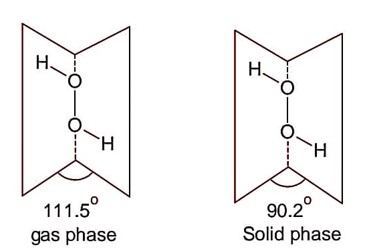
Following are the dihedral angles of H2O2 in gas phase and solid (Reverse with respect to question).
New answer posted
7 months agoContributor-Level 10
In strong field of octahedral complex of Fe2+, the electronic configuration is,
Number of unpaired electrons are zero in the presence of strong field ligand.
Hence, spin only magnetic moment =
New answer posted
7 months agoContributor-Level 10
High leaving tendency corresponds to high reactivity towards hydrolysis. Hence order is.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers