Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoBeginner-Level 5
The name of covalent and ionic bonds tell their formation story in small detail. Co- means sharing or coexisting, so whenever there is bond fromation due to sharing of valence electron, it is known as covalent bond. Similerly, ionic bonds suggest that when a bond formation takes place due to ions, which attract each other, it is considered as ionic bond. Ionic bonds are also known as electrovalent bonds. There are some major differences between Electrovalent and covalent bonds. Chekc the table below:
| Aspect | Covalent Bond | Ionic Bond |
|---|---|---|
| Reason of Formation | sharing of electrons between atoms | transfer of electrons from one atom to another |
| Elements | Usually non-metals with non-metals | Usually metals with non-metals |
| Strength | Moderate | the strongest |
| Nature | Directional Bond & Poor conductors of electricity | Non-directional Bond & Conducts electricity in aqueous state |
| Electronegativity Difference | Small (generally < 1.7) | Large (generally > 1.7) |
| Physical State | Usually Gases, liquids, and soft solids | Hard crystalline solids |
| Melting & Boiling Points | low to moderate | high |
| Solubility | Non-polar solvents | Polar solvents like water |
| Examples | H? , O? , CH? , HCl, NH? | NaCl, KBr, CaCl? , MgO |
For more detail related to the chemical bonding in class 11 chemistry read our notes.
New answer posted
8 months agoContributor-Level 10
Ala – Gly – Leu-Ala- Val
Two amino acid units are connected by peptide linkage. So number of peptide linkage = 4.
New answer posted
8 months agoContributor-Level 10
Number of moles of gas =
Mass of nitrogen =
% of N in organic compound =
New answer posted
8 months agoContributor-Level 10
In acidic solution
Difference in oxidation state of Mn is product
= 7 – 4 = 3
New answer posted
8 months agoContributor-Level 10
Baryte – BaSO4 [Sulphate based ore]
Galena – PbS
Zinc blende – ZnS
Copper pyrite – CuFeS2
New answer posted
8 months agoContributor-Level 10
Number of moles of CH3COOH Adsorbed on 0.6 gm wood charcoal.
= 20 * 10-3 moles
Mass of CH3COOH absorbed = 20 * 10-3 * 60 = 1.2 gm
Mass of CH3COOH adsorbed per gram =
New answer posted
8 months agoContributor-Level 10
Eq.(5 – x) mole x molex mole
Total number of moles at equilibrium
= 1107 * 103 atm
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers

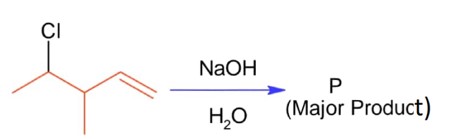
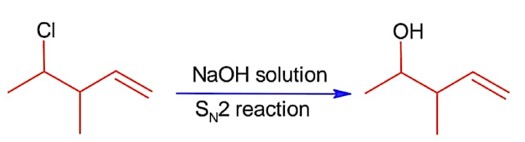 ffffffffffffffffffffffffffffffffffffffffffffffffffffffffffffffffffffffffff
ffffffffffffffffffffffffffffffffffffffffffffffffffffffffffffffffffffffffff