Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
Copolymer is the polymer formed by two or more monomers having multiple bonds.
Buna – S, PHBV and butadiene – styrene are copolymers. Neoprene is homopolymer.
New answer posted
8 months agoContributor-Level 10
In thin layer chromatography, the spots of colourless compounds, which are invisible to eyes can be detected by putting the plate under UV light.
Another detection technique is to place the plate in a covered jar containing I2 (s). Sometimes an appropriate reagent may also be sprayed on the plate, ninhydrin in case of amino acids
New answer posted
8 months agoContributor-Level 10
CN is strong field ligand
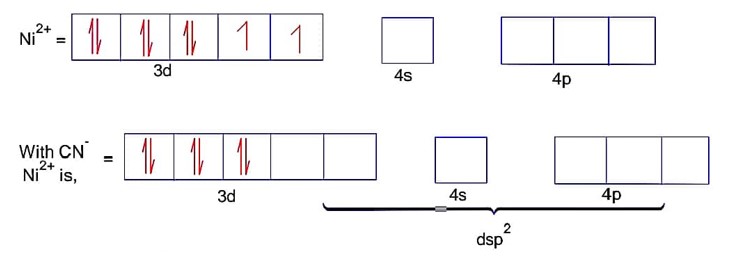
Here; is square planar and diamagnetic.
Ni = 4s23d8Co is strong field ligand.
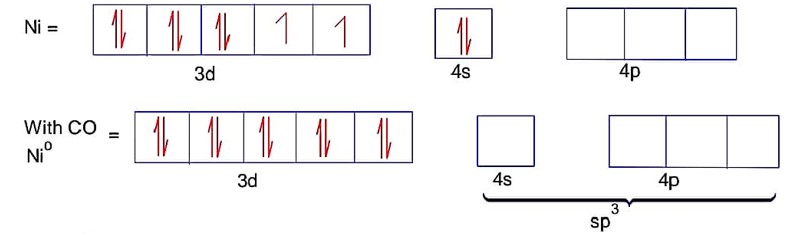
Here ; is tetrahedral and diamagnetic
has 3d8 configuration while has 3d10 configuration.
New answer posted
8 months agoContributor-Level 10
Valence shell electronic configuration of Eu in +2 O.S is,
It has half filled f-sub shell and stable.
New answer posted
8 months agoContributor-Level 10
Pentavalent oxides of group – 15 elements, E2O5 is more acidic than trivalent oxides, E2O3 of the same element.
Acidic strength of trivalent oxides decreases down the group as metallic strength increases.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers