Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
Below 1350° C, Mg can reduce Al2O3 and above 1350°C, Al can reduce MgO (from Ellingham diagram).
Melting and boiling point of Mg are lower than that of Al.
New answer posted
8 months agoContributor-Level 10
General valence shell electronic configuration of group 16 elements is ns2np4. For 3rd period element which is above element “E” is 3s23p4
New answer posted
8 months agoContributor-Level 10
Potential difference between fixed layer and mobile layer of lyophobic colloid is called zeta potential or electro kinetic potential.
This potential difference is related to charge on the surface of colloidal particles.
New answer posted
8 months agoContributor-Level 10
· Density of solid decreases in Schottky defect and vacancy defect.
· Density of solid increases in interstitial defect.
· Density of solid remain unchanged in Frenkel defect.
New answer posted
8 months agoContributor-Level 10
Reaction is
Mass of product, 1, 2, 2- tetrobromopropane obtained
=
= 3.30375
= 3.0375 * 10-1
New answer posted
8 months agoContributor-Level 10
No. of Amino acid units are 4 and
No. of peptide bonds are 3
In tetrapeptide
New answer posted
8 months agoContributor-Level 10
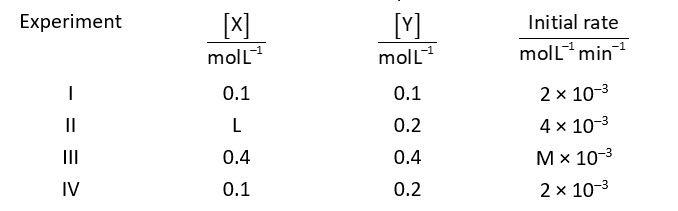
According to condition
Rate
By using experimental data I and II
And by using experimental data I and II
M = 8
Ratio of
New answer posted
8 months agoContributor-Level 10
According to Henry's law
P = KH * mole fraction of solute
Mole of O2 = 1.09 * 10-3
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers