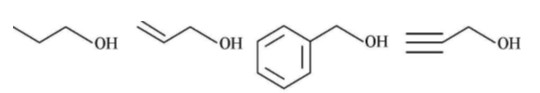
Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
The Pauli exclusion principle states that no two electrons in an atom can have the same four quantum numbers. In other words, only two electrons may exist in the same atomic orbital, and these electrons must have opposite spins. (a) and (f) violate the Pauli exclusion principle. Hund's rule states that the most stable arrangement of electrons in subshells is the one with the greatest number of parallel spins. (b), (d) and (e) violate Hund's rule.
New answer posted
8 months agoContributor-Level 10
Red phosphorus undergoes disproportionation on reaction with alkali to form H4P2O6 and PH3
New answer posted
8 months agoNew answer posted
8 months agoContributor-Level 10
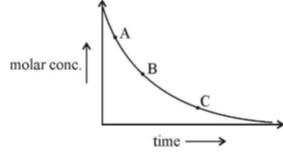
1st order reaction is the fastest at the starting of the reaction then it decrease also you can see the slope which is equal to rate.
New answer posted
8 months agoContributor-Level 10
Kindly consider the following figure
As bond order is increasing it implies bond strength is increasing. As bond order is increasing it implies bond strength is increasing.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 690k Reviews
- 1850k Answers