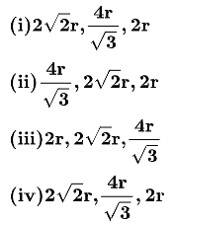Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
53. Options (iii) and (iv) are correct since in anti-ferromagnetic substances the magnetic moments and domains are oppositely oriented, and they cancel out each other's magnetic moment. For example, the anti-ferromagnetic substance-MnO.
New answer posted
10 months agoContributor-Level 10
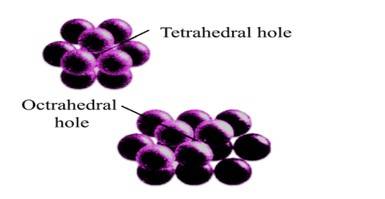
52. Option (iii) and (iv) are correct since tetrahedral voids are formed when the triangular void in the second layer lie exactly above the triangular voids in the first layer and the triangular shape of these voids oppositely overlap while the octahedral voids are formed when triangular void of second layer is not exactly overlap with similar void in first layer. This can be shown as-
New answer posted
10 months agoContributor-Level 10
51. Option (i)? metals >>? insulators < ? semiconductors is correct since metals are the ones which shows the highest conductivity due to the presence of free electrons in them which helps in the conduction of electricity while semiconductors are the ones whose conductivity depends upon the temperature.
Since at normal temperatures, they act as insulators whereas if the temperature rises, the conductivity of the semiconductor also increases. Insulators are the ones which do not have free electrons in them. Due to which they act as poor conductors and hence their conductivity is the lowest.
New answer posted
10 months agoContributor-Level 10
50. Option (i) , ,2r
In FCC crystal - the atoms touch each other at the face diagonals. If a is the edge length of the cube and r is the radius of the atom
Therefore,
a =
In a simple cubic crystal - The atoms at the corners touch each other.
Therefore, a = 2r
New answer posted
10 months agoContributor-Level 10
49. Option (iv) 8 tetrahedral voids within the unit cells is correct since in ccp structure there are 4 atoms per unit cell and the number of tetrahedral voids is twice the number of atoms (i.e. eight tetrahedral voids per unit cell of cubic close packing.
New answer posted
10 months agoContributor-Level 10
48. Option (i) Frenkel defect is correct since in this defect an atom is displaced from its lattice point to an interstitial site thus creating a vacancy at the lattice point. This usually occurs in those ionic crystals where the size of anion is quite large as compared to that of the cation. Here dislocation of atoms occurs and hence it is known as dislocation defect.
New answer posted
10 months agoContributor-Level 10
47. Option (ii) The correct order of the packing efficiency is fcc > bcc > simple cubic
Packing Efficiency (fcc) - 74%
Packing Efficiency (bcc) - 68%
Packing Efficiency (simple cubic) - 52%
New answer posted
10 months agoContributor-Level 10
46. Option (i) all the domains get oriented in the direction of magnetic field is correct since whenever a ferromagnetic substance is placed in a magnetic field it becomes a permanent magnet as all the domains get oriented in the directions of the magnetic field even after removal of the applied magnetic field.
New answer posted
10 months agoContributor-Level 10
45. Option (iv) The fraction of octahedral or tetrahedral voids occupied depends upon the radii of the ions occupying the voids is not true since the occupation of the voids depends upon the stoichiometry of the compound instead of the radii of the ions.
New answer posted
10 months agoContributor-Level 10
44. Option (ii) Ferromagnetic substances cannot be magnetised permanently is correct since ferromagnetic substances are those which are very strongly attracted by the magnetic field and can be magnetized permanently.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers