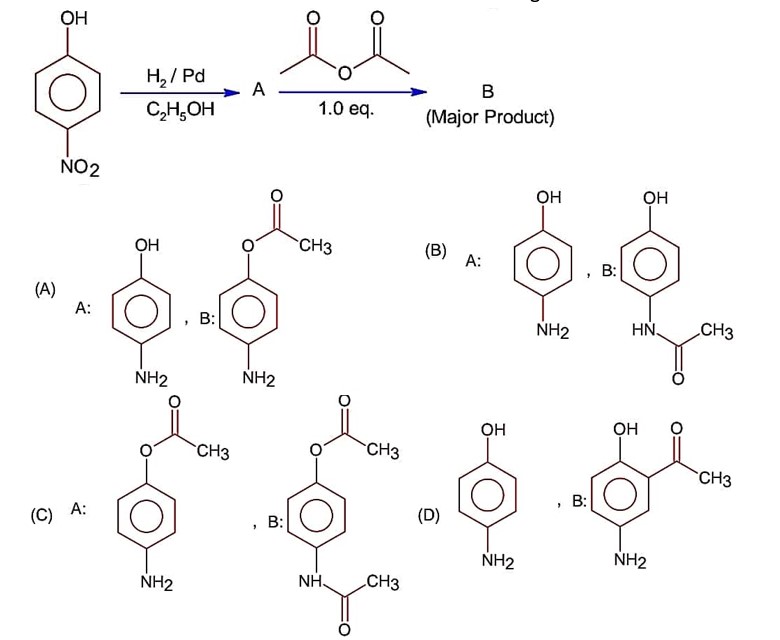
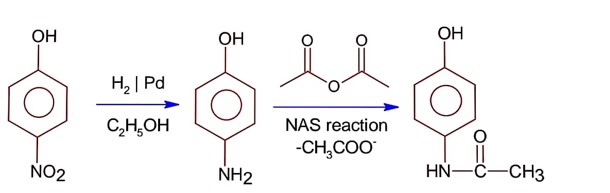
Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
Ozone in the stratosphere is a product of UV radiations acting on O2 molecule.
New answer posted
8 months agoContributor-Level 10
Given that, rate of appearance of Cr2(SO4)3 =
LHS RHS
Rate of disappearance of C2H6O =
Rate of disappearance of C2H6O = 4.005 mol /min
Ans. = 4 (nearest integer)
New answer posted
8 months agoContributor-Level 10
Mass of organic compound = 0.2 gram
= 0.001 mol
Mass of Br = 0.001 * 80g
= 0.08 g
Mass & of Br =
= 40 %
New answer posted
8 months agoContributor-Level 10
Mn6+ has one unpaired electron so paramagnetic and has green colour.
New answer posted
8 months agoContributor-Level 10
MCl3.2L => octahedral complex
It means one Cl- ion present in ionization sphere.
For octahedral complex co-ordination numbers is 6
is bidentate ligand i.e. its denticity is 2.
New answer posted
8 months agoContributor-Level 10
Liquation is applicable for the refining of low melting point metals.
In liquation low melting point metal, melt at first and impurities take more time for melting therefore we can separate metal from their impurities.
New answer posted
8 months agoContributor-Level 10
FeCl3 + K4 [Fe (CN)6] (Potassium ferrocyanide)
excess
|
K Fe [Fe (CN)6]
(colloidal species)
Prussian Blue coloured
colloidal complex
New answer posted
8 months agoContributor-Level 10
(1)
(2)
(3) PbO2 + 2H2O ->Pb (OH)4
(4) SnO2 + 2H2O ->Sn (OH)4
New answer posted
8 months agoContributor-Level 10
MnO -> Antiferromagnetism
O2 -> Paramagnetism
NaCl -> Diamagnetism
Fe3O4 -> Ferrimagnetism
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers