Ncert Solutions Chemistry Class 11th
Get insights from 2k questions on Ncert Solutions Chemistry Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
For spontaneous process
For Isobaric process;
For Isothermal process;
reaction =
New answer posted
8 months agoContributor-Level 10
Density of metal a very specific and depends upon many factors.
Li -> 0.53 gm/cm3
Na -> 0.97 gm/cm3
K -> 0.86 gm/cm3
Rb ->1.53 gm/cm3
Cs -> 1.90 gm/cm3
New answer posted
8 months agoContributor-Level 10
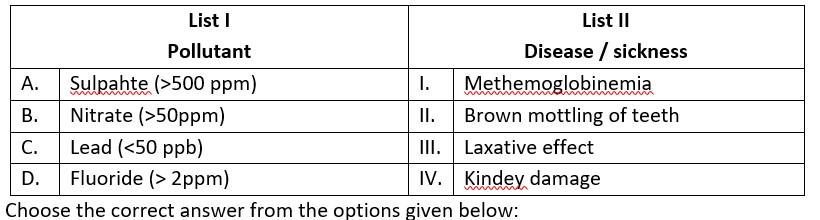
(A) Sulphate (>500 ppm) ® causes laxative effect that leads to dehydration.
(B) Nitrate (> 50 ppm) ® causes methemoglobinemia, skin appears blue.
(C) Lead (>50 ppb) ® It damage kidney and RBC.
(D) Fluoride (>2ppm) ® It causes brown mottling of teeth.
New answer posted
8 months agoContributor-Level 10
Be2Cl4 is Lewis acid and Al2Cl6 has complete octet. Be and Al are amphoteric metal therefore dissolve in acid as well as alkaline solution and form beryllate and aluminate ions in excess alkali.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers