Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
Compounds containing carbonyl
Ligands only are known as homoleptic carbonyl. Such types of compounds are formed by most of the transition metals. These metal carbonyls always have simple, well-defined structures. In metal carbonyls the metal - carbonyl bond possess both s and p.character. M-C-bond is sigma bond. It is formed by the donation of lone pair of electrons of the carbonyl carbon into the vacant orbital of the metal. The M-C pi bond is formed by the donation of a pair of electron from a filled d orbital of a metal into the vacant antibonding? orbital of carbon monoxide. Such type of metal to ligand bonding creates a synergic ef
New answer posted
a year agoContributor-Level 10
5.4
It is important to remove CO (Carbon Monoxide) in the synthesis of ammonia as CO affects the activity of Iron catalyst which is required in Haber's process.
Note: Haber's process is a very important industrial process which is used to produce ammonia.
New answer posted
a year agoContributor-Level 10
The colour of the particular complex compound depends on the crystal field splitting energy (CFSE). This CFSE depends on the nature of the ligand attached to the metal atom. In case of [Fe (CN)6]4– and [Fe (H2O)6]2+ the colour differs due to differences in CFSE .
CN- is a strong field ligand so will have high CFSE than H2O with a low value of CFSE. There is absorption of the energy from the visible region for the d-d transition and corresponding complimentary colour is observed. Thus there is the colour difference.
New answer posted
a year agoContributor-Level 10
In case of [Ni (H2O)6]2+ H2O is a weak field ligand, so it does not cause the pairing of the unpaired electron of Ni2+ ion. Thus there is possibility of the intra d-d transition from the d orbital of lower energy to that of higher energy. Thus the light is absorbed from the visible region and complimentary colour is observed. But in case of [Ni (CN)4]2– CN- is strong field ligand.
Therefore it will cause pairing of the unpaired electrons of Ni2+ ion. There are no unpaired electrons present, so there is no d-d transition and hence it is colourless.
New answer posted
a year agoContributor-Level 10
5.3
As Adsorption is directly proportional to the available surface area and powdered form of a substance have a greater surface area than the crystalline form of the substance.
So, greater the surface area of the adsorbent more is the adsorption.
Hence, powdered substances are more effective adsorbents than their crystalline forms
New answer posted
a year agoContributor-Level 10
Overall charge balance in [Cr (NH3)6]3+ complex:
X + 6 (0) = + 3 X = + 3
Cr is in + 3 oxidation state.
Electronic configuration of Cr in + 2 state: 3d3 . Now ammonia is a weak field ligand so it not causes pairing of the unpaired electron and undergoes hybridisation to form 6 sp3d2 hybrid orbitals filled by the six ammonia ligands. It's geometry is octahedral with unpaired electrons and hence is paramagnetic complex.
In case of [Ni (CN)4]2– ion :
Overall charge balance in [Ni (CN)4]2–complex:
X + 4 (-1) = -2 X = + 2
Ni is in + 2 oxidation state.
Electronic configuration of Ni in + 2 state: 3d8. Now cyanide ion is a strong field ligand so i
New answer posted
a year agoContributor-Level 10
5.2
As Physisorption is Exothermic in nature, which means when gas gets adsorbed on the solid surface, Heat is evolved. So, according to Le-Chatelieres when the temperature is an increased reverse process (Desorption) will be favoured. So, Physisorption decreases with the increase of temperature.
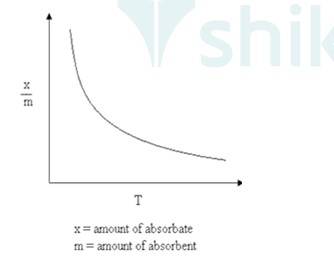
Where x/m: Volume of gas adsorbed
T: Temperature.
New answer posted
a year agoContributor-Level 10
The difference between the energies of the two set of the d orbitals is called as crystal field splitting energy (CFSE). The degenerate d orbitals split into two levels i.e t2g and eg level due to the presence of the ligands. This splitting of the degenerate orbitals due to the ligand is called as crystal field splitting and the energy difference between the two levels is called as crystal field splitting energy.
After the splitting of the degenerate orbitals has taken place the filling of the electrons takes place. Now first 3 electrons goes into the lower energy three t2g orbitals. The fourth electron can be filled in two ways:
It can
New answer posted
a year agoContributor-Level 10
The strong ligands have higher splitting power of d orbitals of the central metal ion, whereas weak ligand has relatively lower splitting power of d orbitals of the central metal ion. The energy difference between t2g and eg sets of d orbitals is CFSE. The strength of the ligands depend on the magnitude of Δ . Strong ligands have larger value of CFSE and in case of weak ligands the CFSE values are smaller. The common ligands can be arranged in a series in the order of their decreasing field strength, as follows.
This series depends on the power of splitting the d orbitals and is called spectrochemical series, The order of field s
New answer posted
a year agoContributor-Level 10
5.1
The two characteristics of Chemisorption are:
1. In Chemisorption which is highly specific in nature, the adsorb ate and adsorbent get attached by chemical bonds which are either covalent or ionic in
2. High activation energy is required and high temperature is also
3. Chemisorption increases with the increase in surface area which results in more number of active
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers
