Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
4.4 Let the reaction be X→Y
As this reaction follows second order kinetics, the rate of the reaction will be,
Rate = k (X)2 → Equation 1
Since the concentration of X is increased three times, Equation 1 will become,
Rate = k (3X)2
= k * 9 (X)2
∴ The rate of formation of Y will become 9 times.
Thus, the rate of formation of Y will become 9 times
New answer posted
a year agoContributor-Level 10
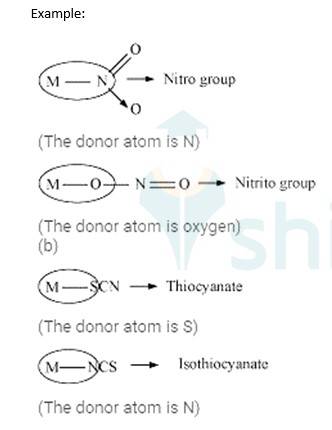
Ligands are the neutral or negatively charged entities surrounding the central metal atom of the coordination complex which possesses at least one unshared pair of electrons.
Based on the number of donor sites of these ligands, Ligands are classified as:
Unidentate ligands: These Ligands which have only one donor site are called unidentate ligands.
Example: F-, Cl – etc.
Didentate ligands: These Ligands which have only two donor site are called didentate ligands.
Example: Ethane-1,2-diamine, Oxalate ion etc.
Ambidentate ligands: These ligands which can attach them with the central metal atom by two different atoms are called as ambidentate
New answer posted
a year agoContributor-Level 10
14.33
The different types of RNA found in the cell are listed below:-
(i) Messenger RNA (m-RNA)
It carries the genetic message code from the DNA to ribosomes. It is produced by the DNA; m-RNA is also single stranded and constitutes about 15% of total RNA.
(ii) Ribosomal RNA (r-RNA)
It is found in the ribosomes and it is usually associated with protein to form the ribosomes. It is synthesised in the nucleus by DNA. It is single stranded, comprising about 80% of total RNA. It is metabolically stable.
(iii) Transfer RNA (t-RNA)
It is synthesised in nucleus by DNA. It is also called soluble RNA. It is single stranded. There are 20
New answer posted
a year agoContributor-Level 10
(1) Coordination Entity: Coordination entity is a charged entity having positive or negative charge in which the central atom is surrounded by molecules which may be neutral/negatively charged called Ligands
Examples:
i. Cationic Complexes: [Cu (H2O)6]2+, [Al (H2O)6]3+
ii. Anionic Complexes: [CuCl4]2-, [Al (H2O)2 (OH)4]-
iii. Neutral Complexes: [Co (NH3)4 Cl2], [Ni (CO)4]
(2) Ligands: Ligands are the neutral or negatively charged entities surrounding the central metal atom of the coordination complex which possesses at least one unshared pair of electrons
Example: F-, Cl-, Br-, I-, H20, and NH3
(3) Coordination Number: Coordina
New answer posted
a year agoContributor-Level 10
14.32
The structural difference between DNA and RNA are as follows:-

The functional difference between DNA and RNA are as follows:-

New answer posted
a year agoContributor-Level 10
The reaction is given below:
FeSO4 + (NH4)2SO4 + 6H2O → FeSO4 (NH4)2SO4.6H2O (Mohr Salt)
FeSO4, when reacted with (NH4)SO4, does not form any complex whereas they form a double salt, FeSO4. (NH4)2SO4.6H2O - (Mohr salt) which dissociates into ions in the solution. So, it gives the test of Fe2+ ions.
CuSO4 + 4NH3 + 5H2O→ [Cu (NH3)4SO4].5H2O
CuSO4 solution when mixed with aqueous ammonia in 1: 4 molar ratio forms a complex with formula [Cu (NH3)]SO4 in which the complex ion, [Cu (NH3)4]2+ does not dissociate to give Cu2+ ions. Therefore, it does not give the tests of the Cu2+ ion.
New answer posted
a year agoContributor-Level 10
Bonding in coordination compounds in terms of Werner's postulates is explained as:
(a) Metals can show two types of valencies which are Primary valency and Secondary
1. Primary Valency: Primary Valency shows Oxidation Primary valencies are ionizable.
2. Secondary Valency: Secondary Valency shows coordination These are non-ionizable.
(b) Both Primary and secondary valency of the metal are to be satisfied which is done by negative ions in case of primary valency and negative or neutral species in case of secondary
(c) Metals have a fixed number of secondary valencies/ Coordination number around the central atom, these secondary
New answer posted
a year agoContributor-Level 10
14.31
In the helical structure of DNA, the two strands are held together by hydrogen bonds between specific pairs of bases.
Cytosine from hydrogen bond with guanine, while adenine forms hydrogen bond with thymine. As a result, the two strands are complementary to each other.
DNA consists of two strands of nucleic acid chains coiled around each other in the form of a double helix. The base of one strand of DNA is paired with bases on other strand by means of hydrogen bonding.
This hydrogen bonding is very specific as the bases can only base pair in a complementary manner.
Adenine pairs with only thymine via 2 hydrogen bonds and guanine pairs
New answer posted
a year agoContributor-Level 10
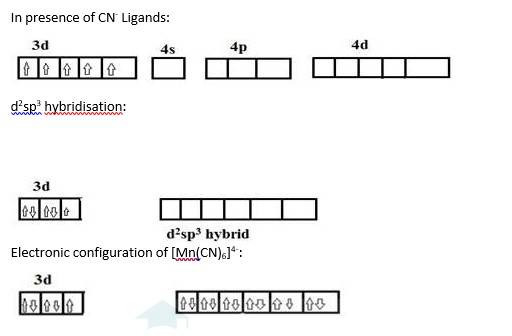
[Mn (H2O)6]+2 | [Mn (CN)6]4- |
Oxidation state of manganese: Overall charge balance: X + 6 (0) = 2 X = + 2 | Oxidation state of manganese: Overall charge balance: X + 6 (-1) = -4 X = + 2 |
Outer electronic configuration of Mn = d5 | Outer electronic configuration of Mn = d5 |
H2O is a weak field ligand so it does not cause pairing of the electron. Therefore Mn undergoes sp3d2 hybridization. Geometry is octahedral. Therefore the 5 unpaired electrons from the d orbital remain as it is. | CN is a strong field ligand so it causes pairing of the electron (5 electrons get paired to form 2 pairs and one unpaired electron). Therefore Mn undergoes d2sp3 hybridization. Geometry is octahedral. |
Mn in + 2 oxidation state:
New answer posted
a year agoContributor-Level 10
In Pt [ (CN)4]2- ion:
Overall charge balance:
X + 4 (-1) = -2 X = + 2.
The oxidation state of Pt is + 2.
Since CN- is a strong field ligand, it causes pairing of the unpaired electrons.
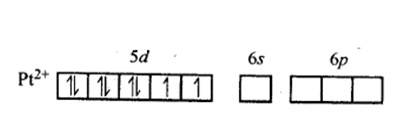
Therefore, now the 2 unpaired electrons from 5d orbital get paired and it undergoes dsp2 hybridisation. It forms square planar geometry. Since all the electrons are paired,
No. of unpaired electrons = 0.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers