Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
14.30
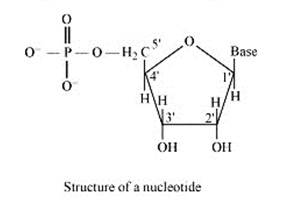
A nucleoside is formed when l-position of a pyrimidine (cytosine, thymine or uracil) or 9- position of a purine (guanine or adenine) base is attached to C-l of sugar (ribose or deoxyribose) by a linkage. Thus in general, nucleosides may be represented as: Sugar-Base.
Nucleoside = sugar + base
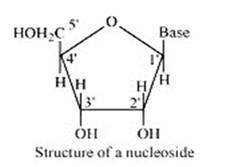
On the other hand, all the three basic components of nucleic acids (i.e., pentose sugar, phosphoric acid and base) are present in a nucleotide.These are obtained by esterification of C5' –OH group of the pentose sugar by phosphoric acid. Thus, in general, a nucleotide is represented as:-
Nucleotide= sugar + base + phosphoric acid
New answer posted
a year agoContributor-Level 10
[Co (NH3)6]3+ | [Ni (NH3)6]2+ |
Oxidation state of cobalt: Overall charge balance: X + 6 (0) = 3 X = + 3 | Oxidation state of nickel: Overall charge balance: X + 6 (0) = 2 X = + 2 |
Outer electronic configuration of cobalt = d6 | Outer electronic configuration of nickel = d8 |
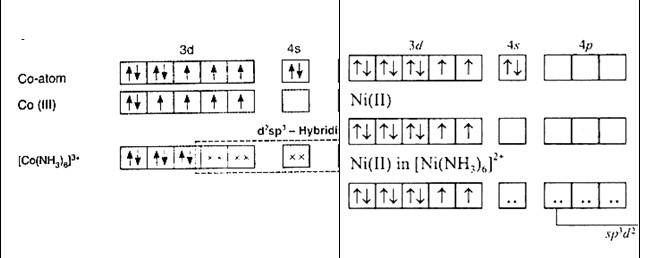
NH3 is a strong field ligand so it causes pairing of electron. Therefore cobalt undergoes d2sp3 hybridisation. As in the hybridisation d2 orbitals are used from the (n-1)d orbitals (inner orbitals as n = 4 being quantum number) . hence it is a inner orbital complex. | NH3 is a strong field ligand so it causes pairing of electron. Therefore, nickel undergoes sp3 d2 hybridisation. As in the hybridisation, d2 orbitals are used from the and orbitals (outer orbitals as n = 4 being quantum number). Hence, it is an outer orbital complex |
New answer posted
a year agoContributor-Level 10
14.29
Nucleic acids are Biomolecules which are found in the nuclei of all living cells, inform of nucleoproteins or chromosomes (proteins containing nucleic acids as the prosthetic group). Nucleic acids are of two types: – deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). Nucleic acids are also known as Polynucleotide as they are long- chain polymers of nucleotides.
The two important functions of nucleic acids are listed below:-
(i) DNA which is responsible for the transference of hereditary effects from one generation to another, which is due to their property of replication during cell division as a result of which two identica
New answer posted
a year agoContributor-Level 10
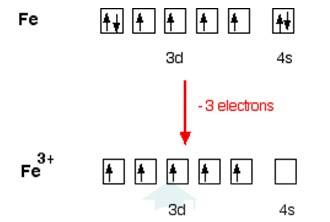
In [Fe (H2O)6]3+
Electronic configuration of Fe is: [Ar]3d64s2
[Ar] = 1s22s22p63s23p6
Electronic configuration of Fe+3 = [Ar]3d5
Outer electronic configuration of Fe+3 = 3d5
Overall charge balance:
X + 6 (0) = 3
X = + 3
In [Fe (CN)6]3-
Overall charge balance:
X + 6 (-1) = -3
X = + 3
In both the compounds Fe is in + 3 oxidation state.
In case of [Fe (H2O)6]3+
H2O is weak field ligand so it does not pair the unpaired electron. Total no. of the unpaired electron, n =5, Spin only magnetic moment is given by:
μ = [n (n + 2)]1/2
μ = [5*7]1/2
μ = 5.916BM
In case of [Fe (CN)6]3-
CN- is a strong field ligand so it pairs up the electron.
Total no. of unpaired e
New answer posted
a year agoContributor-Level 10
14.28
The deficiency of Vitamin A leads to Xerophthalmia which is hardening of the cornea of the eye and night blindness as well. Vitamin-C is essential to us because its deficiency causes scurvy which is a phenomenon of bleeding gums; and pyorrhoea which is phenomenon of loosening-bleeding of teeth.
The sources of vitamin A are fish, cod liver oil, carrots, butter and milk.
The sources of vitamin C are citrus fruits, lemon, amla and green leafy vegetables.
New answer posted
a year agoContributor-Level 10
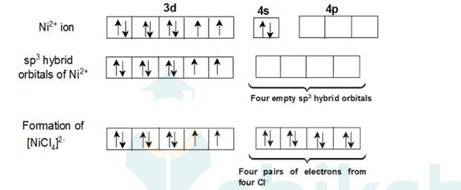
In [Ni (Cl)4]2- ion, Cl- is a weak field ligand so it will not pair the unpaired electrons of Ni+2 ion. Electronic configuration of Ni is: [Ar]3d84s2 where [Ar] = 1s22s22p63s23p6
Electronic configuration of Ni+2 = [Ar]3d8 Outer electronic configuration of Ni+2 = 3d8 Overall charge balance:
X + 4 (-1) = -2 X = + 2.
Therefore it undergoes sp3 hybridization. So it will have tetrahedral geometry.

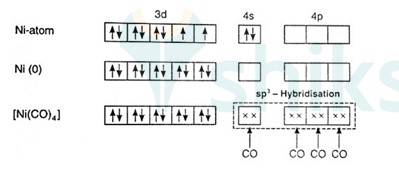
Since there are 2 unpaired electrons in the d orbital so it is a paramagnetic compound. In [Ni (Co)4]:
Overall charge is neutral and oxidation state of Ni can be calculated as:
X + 4 (0) = 0
x = 0
Ni is in zero oxidation state.
Co is a strong field ligand
New answer posted
a year agoContributor-Level 10
According to the valence band theory, the central metal atom or ion under the influence of ligands can use its (n-1)d, ns, np (inner orbital complex) or ns, np, and (outer orbital complex)orbitals for hybridisation to form equivalent set of orbitals of definite geometry.
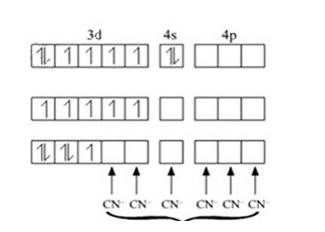
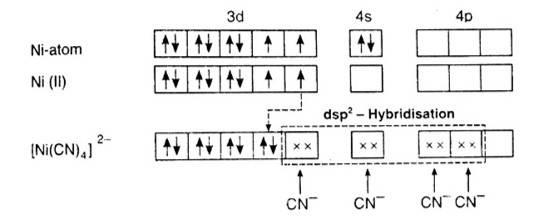
In [Ni (CN)4]2-, oxidation state of Ni can be calculated as :
Using overall charge balance as the whole ion has overall -2 charge:
x + 4 (-1) = -2 (? CN- has -1 negative charge) x = + 2
Ni is in + 2 oxidation state.
Electronic configuration of Ni is: [Ar]3d84s2 Where, [Ar] = 1s22s22p63s23p6
Electronic configuration of Ni+2 = [Ar]3d8 Outer electronic configuration of Ni+2 = 3
New answer posted
a year agoContributor-Level 10
14.27
Vitamins can be defined as the essential dietary factors required by an organism in minute quantities and whose absence causes specific deficiency Vitamins are broadly classified into two types:-
(i) Fat-soluble vitamins:
These are oily substances not readily soluble in water. These include vitamins A, D, E and K. Vitamin K is responsible for the coagulation of blood.
(ii) Water-soluble vitamins:-
Vitamins that are soluble in water belong to this group. For example :B group vitamins (B1, B2, B6, B12, etc.) and vitamin C.
Vitamin H (Biotin) as an exception, it is neither soluble in water nor in fat
New answer posted
a year agoContributor-Level 10
14.26
During denaturation, 2-degree and 3-degree structures of proteins are destroyed but 1- degree structure remains intact. As a result of denaturation, The globular proteins (soluble in water) are converted into Fibrous proteins (insoluble in water) and their biological activity is lost. For example, boiled egg which contains coagulated proteins cannot be hatched to produce chickens.
New answer posted
a year agoContributor-Level 10
These compounds give different ions in aqueous solution. This can be tested by using AgNO3 solution and BaCl2
[Co (NH3)5Cl]SO4 (aq) + BaCl2 (aq) → BaSO4 (ppt)
[Co (NH3)5Cl]SO4 (aq) + AgNO3 (aq) → no reaction
[Co (NH3)5 (SO4)]Cl (aq) + BaCl2 (aq) → no reaction
[Co (NH3)5 (SO4)]Cl (aq) + AgNO3 (aq) → AgCl (ppt)
Hence, they give different precipitates with different solutions. Thus, they are ionisation isomers.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers