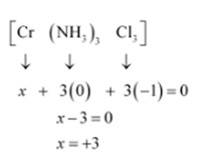Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
(i) No geometrical isomer is possible for [Cr (C2O4)3]3– because the ligand C2O 2- is bidentate ligand (which have two sites of attachment to the central atom) and also in the coordination sphere it is the only ligand bond to it.
(ii) In the coordination sphere of [Co (NH3)3Cl3] there are two types of ligands present i.e NH3 and Cl- . Coordination number is There are 2 isomers possible for the complex:
Facial: In this isomer one type of ligand say NH3 forms the face of the square bipyramidal (triangular) structure.
Meridional: In this isomer one type of ligand are along the central axis of the pyramidal structure.
New answer posted
a year agoContributor-Level 10
4.9 Given-
Energy of activation, Ea= 209.5 kJ mol–1
Ea in joules = 209.5 * 1000 = 209500 J mol–1
Temperature, T = 581K
Gas constant, R = 8.314 J K-1 mol-1
Using the Arrhenius equation,
k = Ae-Ea/RT
In this equation, the term e-Ea/RT represents the number of molecules which have kinetic energy greater than the activation energy, Ea.
∴The number of molecules = e-Ea/RT → Equation 1. Substituting all the known values in equation 1, we get,
= e-209500/8.314X581
=e-43.3708
Finding the value of anti ln (43.3708), we get, 1.47 * 10-19
The fraction of molecules of reactants having energy equal to or greater than activation energy is 1.47 * 10-19
New answer posted
a year ago9.18 List various types of isomerism possible for coordination compounds, giving an example of each.
Contributor-Level 10
Isomers are the compounds which have same chemical formula but different arrangement of atoms in space. There are principle two types of isomerism:
(i) Stereo isomerism
(ii) Geometrical isomerism
(iii) Optical isomerism
(iv) Structural isomerism
(v) Ionisation isomerism
(vi) Linkage isomerism
(vii) Coordination isomerism
(viii) Solvate isomerism
Geometrical isomerism comes into existence by the different spatial arrangement of groups around the central metal atom. Similar groups may either be arranged on the same side or on opposite sides of the central metal atom. This gives rise to two types of isomers called cis and trans isomers. Wh
New answer posted
a year agoContributor-Level 10
4.8 Given-
Initial temperature, T1 = 298 K
Final temperature, T2 = 298 K + 10 K = 308 K
Knowing that the rate constant of a chemical reaction normally increases with increase in temperature, we assume that,
Initial value of rate constant, k1 = k
Final value of rate constant, k2 = 2k
Using Arrhenius equation,

New answer posted
a year agoContributor-Level 10
(i) Starting with cation, the complex ion contains six ammonia molecules with cobalt in + 3 oxidation The name of compound: [Co (NH3)6]Cl3 is hexaamminecobalt (III) chloride.
(ii) The complex ion is cation, so there are 2 ammonia molecules, one chloride ion and methyammine molecule qith platinum in + 2 state. Going in alphabetical order, the name of compound: [Pt (NH3)2Cl (NH2CH3)]Cl is diamminechloridomethylammineplatinum (II)
(iii) It is a complex cation with six water molecules and titanium atom in + 3 state. The name of the compound: [Ti (H2O)6]3+ is hexaaquatitanium (III) ion
(iv) The complex ion is cation with
New answer posted
a year agoContributor-Level 10
4.7 The rate constant of a chemical reaction normally increases with increase in temperature. It is observed that for a chemical reaction with rise in temperature by 10°C, the rate constant is nearly doubled and this temperature dependence of the rate of a chemical reaction can be accurately explained by Arrhenius equation,
k = A
Thus, the rate constant of a chemical reaction normally increases with increase in temperature.
New answer posted
a year agoContributor-Level 10
(i) Tetrahydroxidozincate (II) = [Zn (OH)4]-2
Tetrahydroxi means 4 hydroxide ions with zinc in + 2 oxidation state. Hydroxide ions have a negative charge of -1, so balancing the overall charge of the coordination compound to be zero we get the formula as : [Zn (OH)4]-2
(ii) Potassium tetrachloridopalladate (II) = K3 [PdCl4].
Tetrachlorido means 3 Chloride ions each having a negative charge. Platinum is in + 2 state. Balance overall charge as 0, no. Of potassium ions are 3. Formula: K3 [PdCl4].
(iii) Diamminedichloridoplatinum (II) = [Pt (NH3)Cl2]2+
Diammine means 2 ammonia molecules, dichlorido means 2 chloride ions, platinum
New answer posted
a year agoContributor-Level 10
4.6 Given-
t1/2 = 60 mins
Using the formula for half life, t1/2 = 0.693/k, we get,
k= 0.693 / t1/2
k= 0.693 / 60
∴k = 1.155 * 10-2 min-1
The rate constant of the reaction, k is 1.155 * 10-2 min-1
New answer posted
a year agoContributor-Level 10
(i) Let Oxidation of Co be x and charge on the complex is given as + 2
H2O has Oxidation Number: 0
CN has Oxidation Number: -1
en has Oxidation Number :0
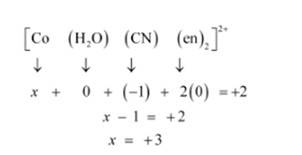
(ii) Let Oxidation number of Co be x and charge on the complex is given as + 1
Br has Oxidation number: 1
en has oxidation number : 0
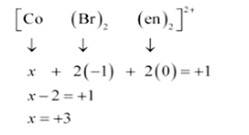
(iii) Let Oxidation number of Pt be x and charge on the complex is given as -2
Cl has oxidation number : -1
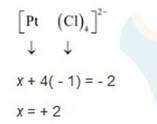
(iv) This complex can also be seen as [Fe (CN)6]3-
Let Oxidation number of Fe be x and charge given on the complex is given as -3
CN has oxidation number : -1
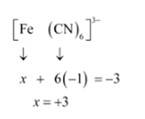
(v) Let Oxidation number of Cr be x and charge given on the complex is given
New answer posted
a year agoContributor-Level 10
4.5 Given-
Rate constant, k = 1.15 * 10-3 s-1
innitial quantity R0 = 5g
Final quantity, R = 3 g
According to the formula of first order reaction
K = 2.303/ t log R0 / R
1.15 X 10-3 = 2.303 /t log 5/3
t = 2.303 / 1.15 X 10-3 log 5/3
t = 443.8 s
t = 4.438 * 102 s
The time taken for 5g of the reactant to reduce to 3 g is 4.438 * 102 s
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers