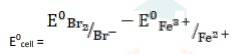Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
14.18
Open structure of D-glucose could not explain the following reactions:
(i) Despite having the aldehyde group, glucose does not give 2,4 DNP test and Schiff's
(ii) Glucose does not react with sodium hydrogen sulphite to form addition
(iii) The absence of free –CHO group is shown when the penta-acetate of glucose does not react with hydroxyl
(iv) When glucose is heated with methanol in the presence of dry HCl gas, it forms two isomeric monomethyl derivatives known as? -D-glucoside and? -D-glucoside. Since only one molecule of methanol is used for the formation of methyl glucosided, these must be hemiacetals.
New answer posted
a year agoContributor-Level 10
14.17
When D-glucose is heated and treated with HI for a long period of time, then n-hexane is formed, which shows that all the six-carbon atoms are linked in a straight
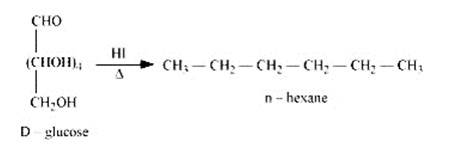
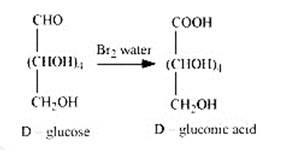
2. When D-glucose is treated with Br2 water i.e., bromine water which is a mild oxidising agent, then we get D-gluconic acid as one of the product. This reaction assures the presence of carbonyl group which is available as an aldehydic group.
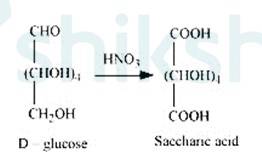
3. On being treated with HNO3, D-glucose get oxidised which gives saccharic acid as final Saccharic acid is a di-carboxylic acid. This reaction confirms the presence of alcoholic group (- OH) in the glucose.
New answer posted
a year agoContributor-Level 10
Given -
(i) All the ions are in aqueous state.
Reaction in solution:
AgNO3 (s) + aq → Ag + + NO3-
H2O → H + + OH -
At cathode:
Ag + (aq) + e - →Ag (s)
Ag + ions have lower discharge potential than H + ions. Hence, Ag + ions get deposited as Ag in preference to H + ions.
At anode:
Ag (s)→ Ag + (aq) + e -
As Ag anode is attacked by NO3- ions, Ag of the anode will dissolve to form Ag + ions in the aqueous solution.
(ii) Reaction in solution:
AgNO3 (s) + aq → Ag + + NO3-
H2O óH + + OH -
At cathode:
2Ag + (aq) + 2e - →2Ag (s)
Ag + ions have lower discharge potential than H + ions. Hence, Ag + ions get deposited as Ag in preference
New answer posted
a year agoContributor-Level 10
The electrode reaction is written as,
2Fe3+ + 2I - → 2Fe2+ + I2
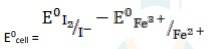
= 0.54V - 0.77V
∴ E0cell = - 0.23 V
It is not feasible, as E0cell is negative, ∴ ?G0 is positive.
- The electrode reaction is written as,
- 2Ag+ (aq) + Cu(s)→ Cu2+ (aq) + Ag(s)
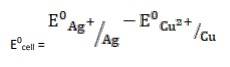
= + 0.80V - 0.34V
∴ E0cell = 0.46V
It is feasible, as Ecell 0 is positive, ∴ ?G0 is negative.
- (iii) The electrode reaction is written as,
- 2Fe3+ (aq) + 2Br- (aq)→ 2Fe2+ (aq) + Br2
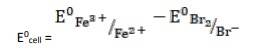 k
k= 0.77V - 1.09V
∴ E0cell = - 0.32 V
It is not feasible, as E0cell is negative, ∴ ?G0 is positive.
- (iv) The electrode reaction is written as,
- Ag(s) + Fe3+ (aq) → Fe2+ (aq) + Ag+ (aq)

= 0.77V - 0.80V
∴ E0cell = -
New answer posted
a year agoContributor-Level 10
14.16
Starch consists of two components – amylase and amylopectin. Amylose is a long linear chain of α–D-(+)-glucose units joined by C1-C4glycosidic linkage (α -link).
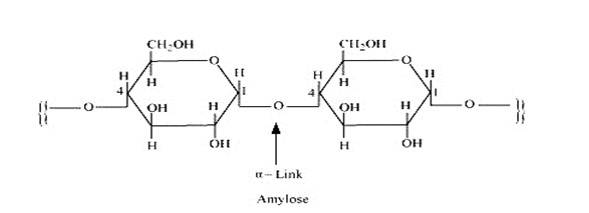
Amylopectin is a branched-chain polymer of –D-glucose units, in which the chain is formed by C1-C4 glycosidic linkage and the branching occurs by C1-C6 glycosidic linkage.

On the other hand, cellulose is the main structural material of tree and other plants. Wood is 50% cellulose, while cotton wool is almost pure cellulose. It is linear chain natural polymers of β-D-glucose units joined by 1, 4-glycosidic linkage (natural linear polymers).

New answer posted
a year agoContributor-Level 10
14.15
Hydrolysis is the process of using water to break down a molecule into two parts. It is usually a type of decomposition reaction where one reactant is water, where water is used to break chemical bonds in the other reactant. It can be considered as reverse of a condensation reaction.
The general formula of a hydrolysis reaction is:
XY + H2O → XH + YOH
(i) On hydrolysis with dilute acids, sucrose yields an equimolecular mixture of α –D glucose and β–D- fructose.
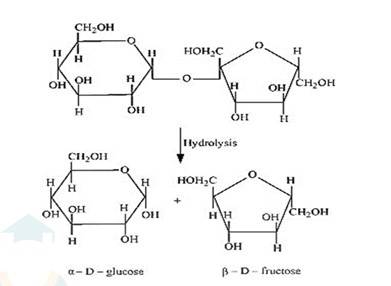
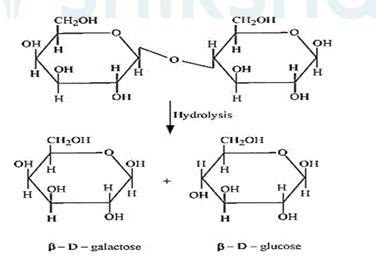
(ii) The hydrolysis of lactose gives β–D-galactose and β–D-glucose as final products.
New answer posted
a year agoContributor-Level 10
Equivalent weight is Ag, EAg = 180/1 = 180
Equivalent weight is Cu, ECu = 63.5 / 2 = 31.75
Equivalent weight is Zn, EZn= 65/2 = 32.5
Using Faraday's second law of electrolysis, to find the mass of Cu and Zn, we use Equation 1,
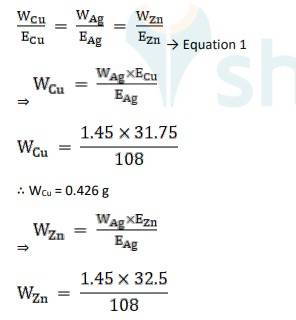
∴ WZn = 0.436 g
To find the time of current flow, using Faraday's first law of electrolysis we get,
M = Z *I *t ⇒ Equation 2
? Z = Equivalent Weight / 96487, Equation 2 becomes,
M = 108 / 96487 X 1.5 X t
t = 1.45 X 96487 / 108X 1.5
t = 864 seconds.
The time of current flow, t = 864 seconds, the mass of Cu is 0.426 g and mass of Zn is 0.436 g
New answer posted
a year agoContributor-Level 10
Quantity of electricity passed = 5 A * (20 * 60 sec)
= 6000 C ⇒ Equation 1
The electrode reaction is written as,
Ni2+ + 2e → Ni
Thus, the quantity of electricity required = 2F
= 2*96487 C
= 192974 C
? 192974 C of electricity deposits 1 mole of Ni, which is 58.7 g ⇒ Equation 2
Thus, equating equations 1 and 2, we get
192974 C of electricity deposits = 58.7 g
6000 C of electricity will deposit = 58.7 X 6000 / 192974
= 1.825g of Ni
The mass of Ni deposited at the cathode is 1.825g of Ni
New answer posted
a year agoContributor-Level 10
(i) The electrode reaction for 1 mole of H2O is given as,
H2O → H2 + 1/2O2
i.e., O2- →1/2 O2 + 2e -
∴ The quantity of electricity required = 2F
= 2*96487 C
= 192974 C
The quantity of electricity required in coulomb for the oxidation of 1 mol of H2O to O2 is 192974 C
(ii) The electrode reaction for 1 mole of FeO is
FeO + 1/2 O2 → 1/2 Fe2O3
i.e., Fe2+ → Fe3+ + e -
∴ The quantity of electricity required = 1F
= 1*96487 C
= 96487 C
The quantity of electricity required in coulomb for the oxidation of 1 mol of FeO to Fe2O3 is 96487 C
New answer posted
a year agoContributor-Level 10
14.14
Glycogen is a polysaccharide-type of carbohydrate. In animals, carbohydrates are stored as glycogen.
But starch is a carbohydrate which consists of two components –amylase (15 -20 %) and amylopectin (80 – 85%). However, glycogen is also like amylopectin but branching will take place after every 5 to 6 glucose unit. Also, glycogen is highly branched.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers